Surface structure and reactivity of
multi-component oxides at the atomic scale
Subproject P02
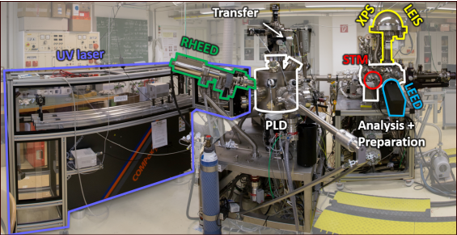
Multi-component metal oxides exhibit a plethora of stoichiometry-dependent structural phases at the surface, even if the composition of the bulk is kept the same. The long-term objective of P02 is to unravel the relationship between surface electronic and geometric structure and reactivity, to ultimately tune these materials for energy-related reactions such as the ORR. The project applies the surface science approach. We will grow well-defined, epitaxial perovskite thin films of LSFO and LSMO in a UHV-based PLD/surface science apparatus under tight control of the surface stoichiometry in the first project period. We will determine the coordinates of surface atoms quantitatively using LEED-IV in close collaboration with theoretical groups.
Theoretical models will also help with interpreting atomically-resolved ncAFM/STM images. These images give direct insights into the behavior of polarons in these complex materials and show how adsorbates such as O2, H2O, CO, and CO2 interact with electronic and structural defects. XPS, TPD, and FTIR of these well-defined systems will deliver desorption energies, vibrational frequencies, and spectral fingerprints. These experimental data on well-defined systems will build a bridge when tested under ‘realistic’ environments at high pressure/temperature and in aqueous solutions. They will also serve to validate ML-based theory approaches.
Expertise
Our expertise is experimental surface science. We operate a total of seven ultrahigh-vacuum (UHV) chambers, which contain virtually all main experimental surface science techniques, as well as an (electro-)chemistry lab.
All chambers are equipped with facilities for sample preparation (sputtering/annealing/gas dosing), as well as various growth techniques (e-beam evaporators, Knudsen cells, UHV-compatible sputter deposition, pulsed laser deposition (PLD)).
Analysis techniques used in our research include:
- Scanning Tunneling Microscopy (STM) (in UHV 4K – 300 K, electrochemical STM)
- Atomic Force Microscopy (AFM): UHV-based (q+ sensor) and in the ambient (cantilever-based)
- Low-Energy Electron Diffraction (LEED)
- Reflection High Energy Diffraction (RHEED)
- X-ray Photoelectron Spectroscopy (XPS)
- Ultraviolet Photoelectron Spectroscopy (UPS)
- Auger Electron Spectroscopy (AES)
- Low-energy He+ ion scattering (LEIS)
- Thermal Programmed Desorption Spectroscopy (TPD)
Team

Former Members
Publications
2023
Raab, Maximilian; Zeininger, Johannes; Suchorski, Yuri; Genest, Alexander; Weigl, Carla; Rupprechter, Günther
Lanthanum modulated reaction pacemakers on a single catalytic nanoparticle
Journal ArticleOpen AccessIn: Nature Communications, vol. 14, no. 7186, 2023.
Abstract | Links | BibTeX | Tags: P08
@article{Raab2023b,
title = {Lanthanum modulated reaction pacemakers on a single catalytic nanoparticle},
author = {Maximilian Raab and Johannes Zeininger and Yuri Suchorski and Alexander Genest and Carla Weigl and Günther Rupprechter},
doi = {10.1038/s41467-023-43026-3},
year = {2023},
date = {2023-11-08},
urldate = {2023-11-08},
journal = {Nature Communications},
volume = {14},
number = {7186},
abstract = {Promoters are important in catalysis, but the atomistic details of their function and particularly their role in reaction instabilities such as kinetic phase transitions and oscillations are often unknown. Employing hydrogen oxidation as probe reaction, a Rh nanotip for mimicking a single Rh nanoparticle and field electron microscopy for in situ monitoring, we demonstrate a La-mediated local catalytic effect. The oscillatory mode of the reaction provides a tool for studying the interplay between different types of reaction pacemakers, i.e., specific local surface atomic configurations that initiate kinetic transitions. The presence of La shifts the bistable reaction states, changes the oscillation pattern and deactivates one of two pacemaker types for the La-free surface. The observed effects originate from the La-enhanced oxygen activation on the catalyst. The experimental observations are corroborated by micro-kinetic model simulations comprising a system of 25 coupled oscillators.},
keywords = {P08},
pubstate = {published},
tppubtype = {article}
}

Tangpakonsab, Parinya; Genest, Alexander; Yang, Jingxia; Meral, Ali; Zou, Bingjie; Yigit, Nevzat; Schwarz, Sabine; Rupprechter, Günther
Kinetic and Computational Studies of CO Oxidation and PROX on Cu/CeO2 Nanospheres
Journal ArticleOpen AccessIn: Topics in Catalysis, vol. 66, pp. 1129–1142, 2023.
Abstract | Links | BibTeX | Tags: P08
@article{Tangpakonsab2023,
title = {Kinetic and Computational Studies of CO Oxidation and PROX on Cu/CeO_{2} Nanospheres},
author = {Parinya Tangpakonsab and Alexander Genest and Jingxia Yang and Ali Meral and Bingjie Zou and Nevzat Yigit and Sabine Schwarz and Günther Rupprechter},
doi = {10.1007/s11244-023-01848-x},
year = {2023},
date = {2023-07-31},
journal = {Topics in Catalysis},
volume = {66},
pages = {1129--1142},
publisher = {Springer Science and Business Media LLC},
abstract = {As supported CuO is well-known for low temperature activity, CuO/CeO_{2} nanosphere catalysts were synthesized and tested for CO oxidation and preferential oxidation of CO (PROX) in excess H_{2}. For the first reaction, ignition was observed at 95 °C, whereas selective PROX occurred in a temperature window from 50 to 100 °C. The catalytic performance was independent of the initial oxidation state of the catalyst (CuO vs. Cu^{0}), suggesting that the same active phase is formed under reaction conditions. Density functional modeling was applied to elucidate the intermediate steps of CO oxidation, as well as those of the comparably less feasible H_{2} transformation. In the simulations, various Cu and vacancy sites were probed as reactive centers enabling specific pathways.},
keywords = {P08},
pubstate = {published},
tppubtype = {article}
}

Asencios, Yvan J. O.; Yigit, Nevzat; Wicht, Thomas; Stöger-Pollach, Michael; Lucrédio, Alessandra F.; Marcos, Francielle C. F.; Assaf, Elisabete M.; Rupprechter, Günther
Partial Oxidation of Bio-methane over Nickel Supported on MgO–ZrO2 Solid Solutions
Journal ArticleOpen AccessIn: Topics in Catalysis, vol. 66, pp. 1539–1552, 2023.
Abstract | Links | BibTeX | Tags: P08
@article{Asencios2023,
title = {Partial Oxidation of Bio-methane over Nickel Supported on MgO–ZrO_{2} Solid Solutions},
author = {Yvan J. O. Asencios and Nevzat Yigit and Thomas Wicht and Michael Stöger-Pollach and Alessandra F. Lucrédio and Francielle C. F. Marcos and Elisabete M. Assaf and Günther Rupprechter},
doi = {10.1007/s11244-023-01822-7},
year = {2023},
date = {2023-05-24},
urldate = {2023-05-24},
journal = {Topics in Catalysis},
volume = {66},
pages = {1539--1552},
publisher = {Springer Science and Business Media LLC},
abstract = {Syngas can be produced from biomethane via Partial Oxidation of Methane (POM), being an attractive route since it is ecofriendly and sustainable. In this work, catalysts of Ni supported on MgO–ZrO_{2} solid solutions, prepared by a one-step polymerization method, were characterized by HRTEM/EDX, XRD, XPS, H_{2}-TPR, and in situ XRD. All catalysts, including Ni/ZrO_{2} and Ni/MgO as reference, were tested for POM (CH_{4}:O_{2} molar ratio 2, 750 ºC, 1 atm). NiO/MgO/ZrO_{2} contained two solid-solutions, MgO–ZrO2 and NiO-MgO, as revealed by XRD and XPS. Ni (30 wt%) supported on MgO–ZrO_{2} solid solution exhibited high methane conversion and hydrogen selectivity. However, depending on the MgO amount (0, 4, 20, 40, 100 molar percent) major differences in NiO reducibility, growth of Ni^{0} crystallite size during H_{2} reduction and POM, and in carbon deposition rates were observed. Interestingly, catalysts with lower MgO content achieved the highest CH_{4} conversion (~ 95%), high selectivity to H_{2} (1.7) and CO (0.8), and low carbon deposition rates (0.024 g_{carbon} g_{cat}^{−1} h^{−1}) with Ni_{4}MgZr (4 mol% MgO) turning out to be the best catalyst. In situ XRD during POM indicated metallic Ni nanoparticles (average crystallite size of 31 nm), supported by MgO–ZrO_{2} solid solution, with small amounts of NiO–MgO being present as well. The presence of MgO also influenced the morphology of the carbon deposits, leading to filaments instead of amorphous carbon. A combustion-reforming mechanism is suggested and using a MgO–ZrO_{2} solid solution support strongly improves catalytic performance, which is attributed to effective O_{2}, CO_{2} and H_{2}O activation at the Ni/MgO–ZrO_{2} interface.},
keywords = {P08},
pubstate = {published},
tppubtype = {article}
}
Winkler, Philipp; Raab, Maximilian; Zeininger, Johannes; Rois, Lea M.; Suchorski, Yuri; Stöger-Pollach, Michael; Amati, Matteo; Parmar, Rahul; Gregoratti, Luca; Rupprechter, Günther
Journal ArticleOpen AccessIn: ACS Catalysis, vol. 13, no. 11, pp. 7650–7660, 2023.
Abstract | Links | BibTeX | Tags: P08
@article{doi:10.1021/acscatal.3c00060,
title = {Imaging Interface and Particle Size Effects by In Situ Correlative Microscopy of a Catalytic Reaction},
author = {Philipp Winkler and Maximilian Raab and Johannes Zeininger and Lea M. Rois and Yuri Suchorski and Michael Stöger-Pollach and Matteo Amati and Rahul Parmar and Luca Gregoratti and Günther Rupprechter},
doi = {10.1021/acscatal.3c00060},
year = {2023},
date = {2023-05-23},
urldate = {2023-01-01},
journal = {ACS Catalysis},
volume = {13},
number = {11},
pages = {7650--7660},
abstract = {The catalytic behavior of Rh particles supported by three different materials (Rh, Au, and ZrO_{2_{) in H_{2_{ oxidation has been studied in situ by correlative photoemission electron microscopy (PEEM) and scanning photoemission electron microscopy (SPEM). Kinetic transitions between the inactive and active steady states were monitored, and self-sustaining oscillations on supported Rh particles were observed. Catalytic performance differed depending on the support and Rh particle size. Oscillations varied from particle size-independent (Rh/Rh) via size-dependent (Rh/ZrO_{2_{) to fully inhibited (Rh/Au). For Rh/Au, the formation of a surface alloy induced such effects, whereas for Rh/ZrO_{2_{, the formation of substoichiometric Zr oxides on the Rh surface, enhanced oxygen bonding, Rh-oxidation, and hydrogen spillover onto the ZrO_{2_{ support were held responsible. The experimental observations were complemented by micro-kinetic simulations, based on variations of hydrogen adsorption and oxygen binding. The results demonstrate how correlative in situ surface microscopy enables linking of the local structure, composition, and catalytic performance.},
keywords = {P08},
pubstate = {published},
tppubtype = {article}
}

Pramhaas, Verena; Unterhalt, Holger; Freund, Hans-Joachim; Rupprechter, Günther
Journal ArticleIn: Angewandte Chemie - International Edition, vol. 62, no. 19, 2023.
Abstract | Links | BibTeX | Tags: P08
@article{Pramhaas2023,
title = {Polarization-Dependent Sum-Frequency-Generation Spectroscopy for In Situ Tracking of Nanoparticle Morphology},
author = {Verena Pramhaas and Holger Unterhalt and Hans-Joachim Freund and Günther Rupprechter},
doi = {10.1002/anie.202300230},
year = {2023},
date = {2023-03-08},
journal = {Angewandte Chemie - International Edition},
volume = {62},
number = {19},
publisher = {Wiley},
abstract = {The surface structure of oxide-supported metal nanoparticles can be determined via characteristic vibrations of adsorbed probe molecules such as CO. Usually, spectroscopic studies focus on peak position and intensity, which are related to binding geometries and number of adsorption sites, respectively. Employing two differently prepared model catalysts, it is demonstrated that polarization-dependent sum-frequency-generation (SFG) spectroscopy reveals the average surface structure and shape of the nanoparticles. SFG results for different particle sizes and morphologies are compared to direct real-space structure analysis by TEM and STM. The described feature of SFG could be used to monitor particle restructuring in situ and may be a valuable tool for operando catalysis.},
keywords = {P08},
pubstate = {published},
tppubtype = {article}
}

Raab, Maximilian; Zeininger, Johannes; Suchorski, Yuri; Tokuda, Keita; Rupprechter, Günther
Emergence of chaos in a compartmentalized catalytic reaction nanosystem
Journal ArticleOpen AccessIn: Nature Communications, vol. 14, pp. 736–745, 2023.
Abstract | Links | BibTeX | Tags: P08
@article{Raab2023,
title = {Emergence of chaos in a compartmentalized catalytic reaction nanosystem},
author = {Maximilian Raab and Johannes Zeininger and Yuri Suchorski and Keita Tokuda and Günther Rupprechter},
doi = {10.1038/s41467-023-36434-y},
year = {2023},
date = {2023-02-10},
urldate = {2023-02-01},
journal = {Nature Communications},
volume = {14},
pages = {736--745},
publisher = {Springer Science and Business Media LLC},
abstract = {In compartmentalized systems, chemical reactions may proceed in differing ways even in adjacent compartments. In compartmentalized nanosystems, the reaction behaviour may deviate from that observed on the macro- or mesoscale. In situ studies of processes in such nanosystems meet severe experimental challenges, often leaving the field to theoretical simulations. Here, a rhodium nanocrystal surface consisting of different nm-sized nanofacets is used as a model of a compartmentalized reaction nanosystem. Using field emission microscopy, different reaction modes are observed, including a transition to spatio-temporal chaos. The transitions between different modes are caused by variations of the hydrogen pressure modifying the strength of diffusive coupling between individual nanofacets. Microkinetic simulations, performed for a network of 52 coupled oscillators, reveal the origins of the different reaction modes. Since diffusive coupling is characteristic for many living and non-living compartmentalized systems, the current findings may be relevant for a wide class of reaction systems.},
keywords = {P08},
pubstate = {published},
tppubtype = {article}
}

Maqbool, Qaisar; Yigit, Nevzat; Stöger-Pollach, Michael; Ruello, Maria Letizia; Tittarelli, Francesca; Rupprechter, Günther
Operando monitoring of a room temperature nanocomposite methanol sensor
Journal ArticleOpen AccessIn: Catalysis Science & Technology, vol. 13, iss. 3, pp. 624–636, 2023.
Abstract | Links | BibTeX | Tags: P08
@article{Maqbool2023,
title = {\textit{Operando} monitoring of a room temperature nanocomposite methanol sensor},
author = {Qaisar Maqbool and Nevzat Yigit and Michael Stöger-Pollach and Maria Letizia Ruello and Francesca Tittarelli and Günther Rupprechter},
doi = {10.1039/d2cy01395a},
year = {2023},
date = {2023-02-07},
urldate = {2023-02-07},
journal = {Catalysis Science & Technology},
volume = {13},
issue = {3},
pages = {624--636},
publisher = {Royal Society of Chemistry (RSC)},
abstract = {The sensing of volatile organic compounds by composites containing metal oxide semiconductors is typically explained via adsorption–desorption and surface electrochemical reactions changing the sensor's resistance. The analysis of molecular processes on chemiresistive gas sensors is often based on indirect evidence, whereas \textit{in situ} or \textit{operando} studies monitoring the gas/surface interactions enable a direct insight. Here we report a cross-disciplinary approach employing spectroscopy of working sensors to investigate room temperature methanol detection, contrasting well-characterized nanocomposite (TiO_{2}@rGO-NC) and reduced-graphene oxide (rGO) sensors. Methanol interactions with the sensors were examined by (quasi) \textit{operando}-DRIFTS and \textit{in situ}-ATR-FTIR spectroscopy, the first paralleled by simultaneous measurements of resistance. The sensing mechanism was also studied by mass spectroscopy (MS), revealing the surface electrochemical reactions. The \textit{operando} and \textit{in situ} spectroscopy techniques demonstrated that the sensing mechanism on the nanocomposite relies on the combined effect of methanol reversible physisorption and irreversible chemisorption, sensor modification over time, and electron/O_{2} depletion–restoration due to a surface electrochemical reaction forming CO_{2} and H_{2}O.},
keywords = {P08},
pubstate = {published},
tppubtype = {article}
}
2022

Zeininger, Johannes; Raab, Maximilian; Suchorski, Yuri; Buhr, Sebastian; Stöger-Pollach, Michael; Bernardi, Johannes; Rupprechter, Günther
Reaction Modes on a Single Catalytic Particle: Nanoscale Imaging and Micro-Kinetic Modeling
Journal ArticleOpen AccessIn: ACS Catalysis, vol. 12, no. 20, pp. 12774–12785, 2022.
Abstract | Links | BibTeX | Tags: P08
@article{Zeininger2022,
title = {Reaction Modes on a Single Catalytic Particle: Nanoscale Imaging and Micro-Kinetic Modeling},
author = {Johannes Zeininger and Maximilian Raab and Yuri Suchorski and Sebastian Buhr and Michael Stöger-Pollach and Johannes Bernardi and Günther Rupprechter},
doi = {10.1021/acscatal.2c02901},
year = {2022},
date = {2022-10-07},
journal = {ACS Catalysis},
volume = {12},
number = {20},
pages = {12774--12785},
publisher = {American Chemical Society (ACS)},
abstract = {The kinetic behavior of individual Rh(\textit{hkl}) nanofacets coupled in a common reaction system was studied using the apex of a curved rhodium microcrystal (radius of 0.65 μm) as a model of a single catalytic particle and field electron microscopy for in situ imaging of catalytic hydrogen oxidation. Depending on the extent of interfacet coupling via hydrogen diffusion, different oscillating reaction modes were observed including highly unusual multifrequential oscillations: differently oriented nanofacets oscillated with differing frequencies despite their immediate neighborhood. The transitions between different modes were induced by variations in the particle temperature, causing local surface reconstructions, which create locally protruding atomic rows. These atomic rows modified the coupling strength between individual nanofacets and caused the transitions between different oscillating modes. Effects such as entrainment, frequency locking, and reconstruction-induced collapse of spatial coupling were observed. To reveal the origin of the different experimentally observed effects, microkinetic simulations were performed for a network of 105 coupled oscillators, modeling the individual nanofacets communicating via hydrogen surface diffusion. The calculated behavior of the oscillators, the local frequencies, and the varying degree of spatial synchronization describe the experimental observations well.},
keywords = {P08},
pubstate = {published},
tppubtype = {article}
}

Zeininger, Johannes; Winkler, Philipp; Raab, Maximilian; Suchorski, Yuri; Prieto, Mauricio J.; Tănase, Liviu C.; Caldas, Lucas Souza; Tiwari, Aarti; Schmidt, Thomas; Stöger-Pollach, Michael; Steiger-Thirsfeld, Andreas; Cuenya, Beatriz Roldan; Rupprechter, Günther
Pattern Formation in Catalytic H2 Oxidation on Rh: Zooming in by Correlative Microscopy
Journal ArticleOpen AccessIn: ACS Catalysis, vol. 12, no. 19, pp. 11974–11983, 2022.
Abstract | Links | BibTeX | Tags: P08
@article{Zeininger2022a,
title = {Pattern Formation in Catalytic H_{2} Oxidation on Rh: Zooming in by Correlative Microscopy},
author = {Johannes Zeininger and Philipp Winkler and Maximilian Raab and Yuri Suchorski and Mauricio J. Prieto and Liviu C. Tănase and Lucas Souza Caldas and Aarti Tiwari and Thomas Schmidt and Michael Stöger-Pollach and Andreas Steiger-Thirsfeld and Beatriz Roldan Cuenya and Günther Rupprechter},
doi = {10.1021/acscatal.2c03692},
year = {2022},
date = {2022-09-19},
urldate = {2022-09-19},
journal = {ACS Catalysis},
volume = {12},
number = {19},
pages = {11974--11983},
publisher = {American Chemical Society (ACS)},
abstract = {Spatio-temporal nonuniformities in H_{2} oxidation on individual Rh(\textit{h k l}) domains of a polycrystalline Rh foil were studied in the 10^{–6} mbar pressure range by photoemission electron microscopy (PEEM), X-ray photoemission electron microscopy (XPEEM), and low-energy electron microscopy (LEEM). The latter two were used for in situ correlative microscopy to zoom in with significantly higher lateral resolution, allowing detection of an unusual island-mediated oxygen front propagation during kinetic transitions. The origin of the island-mediated front propagation was rationalized by model calculations based on a hybrid approach of microkinetic modeling and Monte Carlo simulations.},
keywords = {P08},
pubstate = {published},
tppubtype = {article}
}

Yigit, Nevzat; Genest, Alexander; Terloev, Schamil; Möller, Jury; Rupprechter, Günther
Journal ArticleOpen AccessIn: Journal of Physics: Condensed Matter, vol. 34, no. 35, pp. 354001, 2022.
Abstract | Links | BibTeX | Tags: P08
@article{Yigit2022,
title = {Active sites and deactivation of room temperature CO oxidation on Co_{3}O_{4} catalysts: combined experimental and computational investigations},
author = {Nevzat Yigit and Alexander Genest and Schamil Terloev and Jury Möller and Günther Rupprechter},
doi = {10.1088/1361-648x/ac718b},
year = {2022},
date = {2022-06-29},
urldate = {2022-06-29},
journal = {Journal of Physics: Condensed Matter},
volume = {34},
number = {35},
pages = {354001},
publisher = {IOP Publishing},
abstract = {Co_{3}O_{4} is a well-known low temperature CO oxidation catalyst, but it often suffers from deactivation. We have thus examined room temperature (RT) CO oxidation on Co_{3}O_{4} catalysts by operando DSC, TGA and MS measurements, as well as by pulsed chemisorption to differentiate the contributions of CO adsorption and reaction to CO_{2}. Catalysts pretreated in oxygen at 400 °C are most active, with the initial interaction of CO and Co_{3}O_{4} being strongly exothermic and with maximum amounts of CO adsorption and reaction. The initially high RT activity then levels-off, suggesting that the oxidative pretreatment creates an oxygen-rich reactive Co_{3}O_{4} surface that upon reaction onset loses its most active oxygen. This specific active oxygen is not reestablished by gas phase O_{2} during the RT reaction. When the reaction temperature is increased to 150 °C, full conversion can be maintained for 100 h, and even after cooling back to RT. Apparently, deactivating species are avoided this way, whereas exposing the active surface even briefly to pure CO leads to immediate deactivation. Computational modeling using DFT helped to identify the CO adsorption sites, determine oxygen vacancy formation energies and the origin of deactivation. A new species of CO bonded to oxygen vacancies at RT was identified, which may block a vacancy site from further reaction unless CO is removed at higher temperature. The interaction between oxygen vacancies was found to be small, so that in the active state several lattice oxygen species are available for reaction in parallel.},
keywords = {P08},
pubstate = {published},
tppubtype = {article}
}