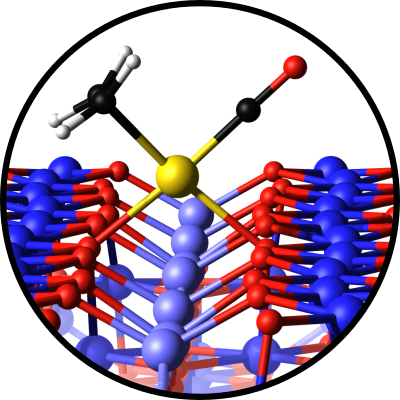
Atomic-scale studies of catalysis
by spinel oxides
Subproject P04
The spinel class of metal oxides hosts diverse materials, some of which make excellent catalysts. Fe3O4 is already the industrial catalyst for the high-temperature water-gas shift reaction (CO+H2O -> H2+CO2), but research is needed to identify the optimal replacement for the toxic Cr promoter/stabilizer. Ternary MeFe2O4 compounds (Me=Fe, Ni, Co, Mn) are active and stable for the electrochemical oxygen evolution reaction (OER). However, the structure of the active catalyst and the reaction mechanisms are unknown. While these reactions appear different, both clearly benefit from a combination of multivalent cations in the surface layers.
In this project, we will seek to learn why, using a combination of atomic-scale imaging, a host of spectroscopies, and theory. We will dope the Fe3O4(001) surface with 3d transition metals and investigate how the adsorption energies, XPS binding energies, and IRAS frequencies of H2O, CO, CO2, O2, and H2 change with sample composition all the way from isolated dopants to ternary thin films. We will use the data obtained in tightly-controlled UHV experiments to:
i) Interpret the reactivity of our model catalysts under realistic HTWGS and OER conditions.
ii) Provide the benchmark data for experiments on nominally similar powder catalysts (P10 Föttinger).
iii) Support the development of theoretical modeling (P07 Franchini).
A joint postdoc (P04-P11) will facilitate the new collaboration with P11 Backus.
Expertise
- Scanning Tunneling Microscopy (STM) (in UHV 4K – 300 K, electrochemical STM)
- Atomic Force Microscopy (AFM): UHV-based (q+ sensor) and in the ambient (cantilever-based)
- Low-Energy Electron Diffraction (LEED)
- Reflection High Energy Diffraction (RHEED)
- X-ray Photoelectron Spectroscopy (XPS)
- Ultraviolet Photoelectron Spectroscopy (UPS)
- Auger Electron Spectroscopy (AES)
- Low-energy He+ ion scattering (LEIS)
- Thermal Programmed Desorption Spectroscopy (TPD)
Team

Associates
Publications
2024
Rath, David; Mikerásek, Vojtěch; Wang, Chunlei; Eder, Moritz; Schmid, Michael; Diebold, Ulrike; Parkinson, Gareth S.; Pavelec, Jiří
Journal ArticleOpen AccessSubmittedarXivIn: arXiv, 2024.
Abstract | Links | BibTeX | Tags: P02, P04
@article{Rath2024,
title = {Infrared Reflection Absorption Spectroscopy Setup with Incidence Angle Selection for Surfaces of Non-Metals},
author = {David Rath and Vojtěch Mikerásek and Chunlei Wang and Moritz Eder and Michael Schmid and Ulrike Diebold and Gareth S. Parkinson and Jiří Pavelec},
url = {https://arxiv.org/abs/2403.19263},
doi = {10.48550/ARXIV.2403.19263},
year = {2024},
date = {2024-03-28},
journal = {arXiv},
publisher = {arXiv},
abstract = {Infrared Reflection Absorption Spectroscopy (IRAS) on dielectric single crystals is challenging because the optimal incidence angles for light-adsorbate interaction coincide with regions of low IR reflectivity. Here, we introduce an optimized IRAS setup that maximizes the signal-to-noise ratio for non-metals. This is achieved by maximizing light throughput, and by selecting optimal incidence angles that directly impact the peak heights in the spectra. The setup uses a commercial FTIR spectrometer and is usable in ultra-high vacuum (UHV). Specifically, the design features sample illumination and collection mirrors with a high numerical aperture inside the UHV system, and an adjustable aperture to select the incidence angle range on the sample. This is important for p-polarized measurements on dielectrics, because the peaks in the spectra reverse direction at the Brewster angle (band inversion). The system components are connected precisely via a single flange, ensuring long-term stability. We studied the signal-to-noise (SNR) variation in p-polarized IRAS spectra for one monolayer of CO on TiO_{2}(110) as a function of incidence angle range, where a maximum signal-to-noise ratio of 70 was achieved at 4 cm^{-1} resolution in five minutes measurement time. The capabilities for s-polarization are demonstrated by measuring one monolayer D_{2}O adsorbed on a TiO_{2}(110) surface, where a SNR of 65 was achieved at a delta_R/R0 peak height of 1.4x10-4 in twenty minutes.},
keywords = {P02, P04},
pubstate = {published},
tppubtype = {article}
}
Wang, Chunlei; Sombut, Panukorn; Puntscher, Lena; Jakub, Zdenek; Meier, Matthias; Pavelec, Jiri; Bliem, Roland; Schmid, Michael; Diebold, Ulrike; Franchini, Cesare; Parkinson, Gareth S.
CO‐Induced Dimer Decay Responsible for Gem‐Dicarbonyl Formation on a Model Single‐Atom Catalyst
Journal ArticleOpen AccessIn PressIn: Angewandte Chemie - International Edition, no. e202317347, 2024, ISSN: 1521-3773.
Abstract | Links | BibTeX | Tags: P02, P04, P07
@article{Wang2024,
title = {CO‐Induced Dimer Decay Responsible for Gem‐Dicarbonyl Formation on a Model Single‐Atom Catalyst},
author = {Chunlei Wang and Panukorn Sombut and Lena Puntscher and Zdenek Jakub and Matthias Meier and Jiri Pavelec and Roland Bliem and Michael Schmid and Ulrike Diebold and Cesare Franchini and Gareth S. Parkinson},
doi = {10.1002/anie.202317347},
issn = {1521-3773},
year = {2024},
date = {2024-01-31},
journal = {Angewandte Chemie - International Edition},
number = {e202317347},
publisher = {Wiley},
abstract = {The ability to coordinate multiple reactants at the same active site is important for the wide-spread applicability of single-atom catalysis. Model catalysts are ideal to investigate the link between active site geometry and reactant binding, because the structure of single-crystal surfaces can be precisely determined, the adsorbates imaged by scanning tunneling microscopy (STM), and direct comparisons made to density functional theory. In this study, we follow the evolution of Rh_{1} adatoms and minority Rh_{2} dimers on Fe_{3}O_{4}(001) during exposure to CO using time-lapse STM at room temperature. CO adsorption at Rh_{1} sites results exclusively in stable Rh_{1}CO monocarbonyls, because the Rh atom adapts its coordination to create a stable pseudo-square planar environment. Rh_{1}(CO)_{2} gem-dicarbonyl species are also observed, but these form exclusively through the breakup of Rh_{2} dimers via an unstable Rh_{2}(CO)_{3} intermediate. Overall, our results illustrate how minority species invisible to area-averaging spectra can play an important role in catalytic systems, and show that the decomposition of dimers or small clusters can be an avenue to produce reactive, metastable configurations in single-atom catalysis.},
keywords = {P02, P04, P07},
pubstate = {published},
tppubtype = {article}
}
2023
Gamba, Oscar; Eder, Moritz; Poglitsch, Matthias; Pavelec, Jiri; Sombut, Panukorn; Meier, Matthias; Diebold, Ulrike; Schmid, Michael; Parkinson, Gareth S.
Formation and stability of Fe-rich terminations of the Fe3O4(001) surface
Journal ArticleOpen AccessIn: Materials Research Express, vol. 10, iss. 44, no. 116517, 2023.
Abstract | Links | BibTeX | Tags: P02, P04
@article{Gamba2023,
title = {Formation and stability of Fe-rich terminations of the Fe_{3}O_{4}(001) surface},
author = {Oscar Gamba and Moritz Eder and Matthias Poglitsch and Jiri Pavelec and Panukorn Sombut and Matthias Meier and Ulrike Diebold and Michael Schmid and Gareth S. Parkinson},
doi = {10.1088/2053-1591/ad0ac5},
year = {2023},
date = {2023-11-22},
urldate = {2023-11-22},
journal = {Materials Research Express},
volume = {10},
number = {116517},
issue = {44},
publisher = {IOP Publishing},
abstract = {Understanding how the structure of iron oxide surfaces varies with their environment is essential for rationalizing their role in (geo-)chemistry and optimizing their application in modern technologies. In this paper, we create Fe-rich terminations of Fe_{3}O_{4}(001) by depositing iron directly onto the 'subsurface cation vacancy'-reconstructed surface, which is the most stable surface under ultrahigh vacuum conditions. Scanning tunneling microscopy and x-ray photoelectron spectroscopy data reveal that the excess iron is initially accommodated as two-fold coordinated adatoms and later incorporates into the subsurface cation vacancies. As the coverage increases, small patches of the octahedral pair termination (also known as the 'Fe dimer' termination) nucleate, eventually covering the entire surface after the deposition of 2 iron atoms per (√2×√2)R45° unit cell. This conclusion effectively rules out some existing models for the termination and provides support for the model proposed by Rustad \textit{et al.} (Surface Science 432, L583-L588, 1999), highlighting the need for further theoretical work to complete the Fe_{3}O_{4}(001) surface phase diagram. The octahedral pair termination is found to be unstable above 523 K and upon exposure to molecular O2 because the excess iron atoms agglomerate to form small FeO_{x} clusters.},
keywords = {P02, P04},
pubstate = {published},
tppubtype = {article}
}
Puntscher, Lena; Sombut, Panukorn; Wang, Chunlei; Ulreich, Manuel; Pavelec, Jiri; Rafsanjani-Abbasi, Ali; Meier, Matthias; Lagin, Adam; Setvin, Martin; Diebold, Ulrike; Franchini, Cesare; Schmid, Michael; Parkinson, Gareth S.
A Multitechnique Study of C2H4 Adsorption on Fe3O4(001)
Journal ArticleOpen AccessIn: Journal of Physical Chemistry C, vol. 127, iss. 37, pp. 18378–18388, 2023.
Abstract | Links | BibTeX | Tags: P02, P04, P07
@article{Puntscher2023,
title = {A Multitechnique Study of C_{2}H_{4} Adsorption on Fe_{3}O_{4}(001)},
author = {Lena Puntscher and Panukorn Sombut and Chunlei Wang and Manuel Ulreich and Jiri Pavelec and Ali Rafsanjani-Abbasi and Matthias Meier and Adam Lagin and Martin Setvin and Ulrike Diebold and Cesare Franchini and Michael Schmid and Gareth S. Parkinson},
doi = {10.1021/acs.jpcc.3c03684},
year = {2023},
date = {2023-09-11},
urldate = {2023-09-11},
journal = {Journal of Physical Chemistry C},
volume = {127},
issue = {37},
pages = {18378--18388},
publisher = {American Chemical Society (ACS)},
abstract = {The adsorption/desorption of ethene (C_{2}H_{4}), also commonly known as ethylene, on Fe_{3}O_{4}(001) was studied under ultrahigh vacuum conditions using temperature-programmed desorption (TPD), scanning tunneling microscopy, X-ray photoelectron spectroscopy, and density functional theory (DFT)-based computations. To interpret the TPD data, we have employed a new analysis method based on equilibrium thermodynamics. C_{2}H_{4} adsorbs intact at all coverages and interacts most strongly with surface defects such as antiphase domain boundaries and Fe adatoms. On the regular surface, C_{2}H_{4} binds atop surface Fe sites up to a coverage of 2 molecules per (√2 × √2)R45° unit cell, with every second Fe occupied. A desorption energy of 0.36 eV is determined by analysis of the TPD spectra at this coverage, which is approximately 0.1–0.2 eV lower than the value calculated by DFT + U with van der Waals corrections. Additional molecules are accommodated in between the Fe rows. These are stabilized by attractive interactions with the molecules adsorbed at Fe sites. The total capacity of the surface for C_{2}H_{4} adsorption is found to be close to 4 molecules per (√2 × √2)R45° unit cell.},
keywords = {P02, P04, P07},
pubstate = {published},
tppubtype = {article}
}
Redondo, Jesús; Michalička, Jan; Kraushofer, Florian; Franceschi, Giada; Šmid, Břetislav; Kumar, Nishant; Man, Ondřej; Blatnik, Matthias; Wrana, Dominik; Mallada, Benjamin; Švec, Martin; Parkinson, Gareth S.; Setvin, Martin; Riva, Michele; Diebold, Ulrike; Čechal, Jan
Journal ArticleOpen AccessIn: Advanced Materials Interfaces, no. 2300602, 2023.
Abstract | Links | BibTeX | Tags: P02, P04
@article{Redondo2023,
title = {Hematite α-Fe_{2}O_{3}(0001) in Top and Side View: Resolving Long-Standing Controversies about Its Surface Structure},
author = {Jesús Redondo and Jan Michalička and Florian Kraushofer and Giada Franceschi and Břetislav Šmid and Nishant Kumar and Ondřej Man and Matthias Blatnik and Dominik Wrana and Benjamin Mallada and Martin Švec and Gareth S. Parkinson and Martin Setvin and Michele Riva and Ulrike Diebold and Jan Čechal},
doi = {10.1002/admi.202300602},
year = {2023},
date = {2023-08-18},
urldate = {2023-08-18},
journal = {Advanced Materials Interfaces},
number = {2300602},
publisher = {Wiley},
abstract = {Hematite is a common iron oxide found in nature, and the α-Fe_{2}O_{3}(0001) plane is prevalent on the nanomaterial utilized in photo- and electrocatalytic applications. The atomic-scale structure of the surface remains controversial despite decades of study, partly because it depends on sample history as well as the preparation conditions. Here, a comprehensive study is performed using an arsenal of surface techniques (non-contact atomic force microscopy, scanning tunneling microscopy, low-energy electron diffraction, and X-ray photoemission spectroscopy) complemented by analyses of the near surface region by high-resolution transmission electron microscopy and electron energy loss spectroscopy. The results show that the so-called “bi-phase” termination forms even under highly oxidizing conditions; a (1 × 1) surface is only observed in the presence of impurities. Furthermore, it is shown that the biphase is actually a continuous layer distorted due to a mismatch with the subsurface layers, and thus not the proposed mixture of FeO(111) and α-Fe_{2}O_{3}(0001) phases. Overall, the results show how combining surface and cross-sectional imaging provides a full view that can be essential for understanding the role of the near-surface region on oxide surface properties.},
keywords = {P02, P04},
pubstate = {published},
tppubtype = {article}
}
Redondo, Jesus; Reticcioli, Michele; Gabriel, Vit; Wrana, Dominik; Ellinger, Florian; Riva, Michele; Franceschi, Giada; Rheinfrank, Erik; Sokolovic, Igor; Jakub, Zdenek; Kraushofer, Florian; Alexander, Aji; Patera, Laerte L.; Repp, Jascha; Schmid, Michael; Diebold, Ulrike; Parkinson, Gareth S.; Franchini, Cesare; Kocan, Pavel; Setvin, Martin
Real-space investigation of polarons in hematite Fe2O3
Journal ArticleSubmittedarXivIn: arXiv, 2023.
Abstract | Links | BibTeX | Tags: P02, P04, P07
@article{Redondo2024,
title = {Real-space investigation of polarons in hematite Fe2O3},
author = {Jesus Redondo and Michele Reticcioli and Vit Gabriel and Dominik Wrana and Florian Ellinger and Michele Riva and Giada Franceschi and Erik Rheinfrank and Igor Sokolovic and Zdenek Jakub and Florian Kraushofer and Aji Alexander and Laerte L. Patera and Jascha Repp and Michael Schmid and Ulrike Diebold and Gareth S. Parkinson and Cesare Franchini and Pavel Kocan and Martin Setvin},
url = {https://arxiv.org/abs/2303.17945},
year = {2023},
date = {2023-03-31},
urldate = {2023-03-31},
journal = {arXiv},
abstract = {In polarizable materials, electronic charge carriers interact with the surrounding ions, leading to quasiparticle behaviour. The resulting polarons play a central role in many materials properties including electrical transport, optical properties, surface reactivity and magnetoresistance, and polaron properties are typically investigated indirectly through such macroscopic characteristics. Here, noncontact atomic force microscopy (nc-AFM) is used to directly image polarons in Fe_{2}O_{3} at the single quasiparticle limit. A combination of Kelvin probe force microscopy (KPFM) and kinetic Monte Carlo (KMC) simulations shows that Ti doping dramatically enhances the mobility of electron polarons, and density functional theory (DFT) calculations indicate that a metallic transition state is responsible for the enhancement. In contrast, hole polarons are significantly less mobile and their hopping is hampered further by the introduction of trapping centres.},
keywords = {P02, P04, P07},
pubstate = {published},
tppubtype = {article}
}

Kraushofer, Florian; Meier, Matthias; Jakub, Zdeněk; Hütner, Johanna; Balajka, Jan; Hulva, Jan; Schmid, Michael; Franchini, Cesare; Diebold, Ulrike; Parkinson, Gareth S.
Oxygen-Terminated (1 × 1) Reconstruction of Reduced Magnetite Fe3O4(111)
Journal ArticleOpen AccessIn: vol. 14, no. 13, pp. 3258–3265, 2023.
Abstract | Links | BibTeX | Tags: P02, P04, P07
@article{Kraushofer2023,
title = {Oxygen-Terminated (1 × 1) Reconstruction of Reduced Magnetite Fe_{3}O_{4}(111)},
author = {Florian Kraushofer and Matthias Meier and Zdeněk Jakub and Johanna Hütner and Jan Balajka and Jan Hulva and Michael Schmid and Cesare Franchini and Ulrike Diebold and Gareth S. Parkinson},
doi = {10.1021/acs.jpclett.3c00281},
year = {2023},
date = {2023-03-28},
urldate = {2023-03-28},
volume = {14},
number = {13},
pages = {3258--3265},
publisher = {American Chemical Society (ACS)},
abstract = {The (111) facet of magnetite (Fe_{3}O_{4}) has been studied extensively by experimental and theoretical methods, but controversy remains regarding the structure of its low-energy surface terminations. Using density functional theory (DFT) computations, we demonstrate three reconstructions that are more favorable than the accepted Feoct2 termination under reducing conditions. All three structures change the coordination of iron in the kagome Feoct1 layer to be tetrahedral. With atomically resolved microscopy techniques, we show that the termination that coexists with the Fetet1 termination consists of tetrahedral iron capped by 3-fold coordinated oxygen atoms. This structure explains the inert nature of the reduced patches.},
keywords = {P02, P04, P07},
pubstate = {published},
tppubtype = {article}
}
2022
Schmid, Michael; Parkinson, Gareth S.; Diebold, Ulrike
Analysis of Temperature-Programmed Desorption via Equilibrium Thermodynamics
Journal ArticleOpen AccessIn: ACS Physical Chemistry Au, vol. 3, iss. 1, pp. 44–62, 2022.
Abstract | Links | BibTeX | Tags: P02, P04
@article{Schmid2022,
title = {Analysis of Temperature-Programmed Desorption via Equilibrium Thermodynamics},
author = {Michael Schmid and Gareth S. Parkinson and Ulrike Diebold},
doi = {10.1021/acsphyschemau.2c00031},
year = {2022},
date = {2022-11-15},
journal = {ACS Physical Chemistry Au},
volume = {3},
issue = {1},
pages = {44--62},
publisher = {American Chemical Society (ACS)},
abstract = {Temperature-programmed desorption (TPD) experiments in surface science are usually analyzed using the Polanyi–Wigner equation and/or transition-state theory. These methods are far from straightforward, and the determination of the pre-exponential factor is often problematic. We present a different method based on equilibrium thermodynamics, which builds on an approach previously used for TPD by Kreuzer et al. (\textit{Surf. Sci.}\textbf{1988}). Equations for the desorption rate are presented for three different types of surface–adsorbate interactions: (i) a 2D ideal hard-sphere gas with a negligible diffusion barrier, (ii) an ideal lattice gas, that is, fixed adsorption sites without interaction between the adsorbates, and (iii) a lattice gas with a distribution of (site-dependent) adsorption energies. We show that the coverage dependence of the sticking coefficient for adsorption at the desorption temperature determines whether the desorption process can be described by first- or second-order kinetics. The sticking coefficient at the desorption temperature must also be known for a quantitative determination of the adsorption energy, but it has a rather weak influence (like the pre-exponential factor in a traditional TPD analysis). Quantitative analysis is also influenced by the vibrational contributions to the energy and entropy. For the case of a single adsorption energy, we provide equations to directly convert peak temperatures into adsorption energies. These equations also provide an approximation of the desorption energy in cases that cannot be described by a fixed pre-exponential factor. For the case of a distribution of adsorption energies, the desorption spectra cannot be considered a superposition of desorption spectra corresponding to the different energies. Nevertheless, we present a method to extract the distribution of adsorption energies from TPD spectra, and we rationalize the energy resolution of TPD experiments. The analytical results are complemented by a program for simulation and analysis of TPD data.},
keywords = {P02, P04},
pubstate = {published},
tppubtype = {article}
}
Wang, Zhichang; Reticcioli, Michele; Jakub, Zdenek; Sokolović, Igor; Meier, Matthias; Boatner, Lynn A; Schmid, Michael; Parkinson, Gareth S.; Diebold, Ulrike; Franchini, Cesare; Setvin, Martin
Surface chemistry on a polarizable surface: Coupling of CO with KTaO 3(001)
Journal ArticleOpen AccessIn: Science Advances, vol. 8, iss. 33, 2022.
Abstract | Links | BibTeX | Tags: P02, P04, P07
@article{Wang2022,
title = {Surface chemistry on a polarizable surface: Coupling of CO with KTaO _{3}(001)},
author = {Zhichang Wang and Michele Reticcioli and Zdenek Jakub and Igor Sokolović and Matthias Meier and Lynn A Boatner and Michael Schmid and Gareth S. Parkinson and Ulrike Diebold and Cesare Franchini and Martin Setvin},
url = {https://www.science.org/doi/10.1126/sciadv.abq1433},
doi = {10.1126/sciadv.abq1433},
year = {2022},
date = {2022-08-19},
urldate = {2022-08-19},
journal = {Science Advances},
volume = {8},
issue = {33},
publisher = {American Association for the Advancement of Science (AAAS)},
abstract = {Polarizable materials attract attention in catalysis because they have a free parameter for tuning chemical reactivity. Their surfaces entangle the dielectric polarization with surface polarity, excess charge, and orbital hybridization. How this affects individual adsorbed molecules is shown for the incipient ferroelectric perovskite KTaO_{3}. This intrinsically polar material cleaves along (001) into KO- and TaO_{2}-terminated surface domains. At TaO_{2} terraces, the polarity-compensating excess electrons form a two-dimensional electron gas and can also localize by coupling to ferroelectric distortions. TaO_{2} terraces host two distinct types of CO molecules, adsorbed at equivalent lattice sites but charged differently as seen in atomic force microscopy/scanning tunneling microscopy. Temperature-programmed desorption shows substantially stronger binding of the charged CO; in density functional theory calculations, the excess charge favors a bipolaronic configuration coupled to the CO. These results pinpoint how adsorption states couple to ferroelectric polarization.},
keywords = {P02, P04, P07},
pubstate = {published},
tppubtype = {article}
}
![Role of Polarons in Single-Atom Catalysts: Case Study of Me1[Au1,Pt1 and Rh1] on TiO2(110)](https://sfb-taco.at/wp-content/uploads/2023/02/P07_P04-300x300.png)
Sombut, Panukorn; Puntscher, Lena; Atzmüller, Marlene; Jakub, Zdenek; Reticcioli, Michele; Meier, Matthias; Parkinson, Gareth S.; Franchini, Cesare
Role of Polarons in Single-Atom Catalysts: Case Study of Me1[Au1,Pt1 and Rh1] on TiO2(110)
Journal ArticleOpen AccessIn: Topics in Catalysis, vol. 65, pp. 1620–1630, 2022.
Abstract | Links | BibTeX | Tags: P04, P07
@article{Sombut2022,
title = {Role of Polarons in Single-Atom Catalysts: Case Study of Me_{1}[Au_{1},Pt_{1} and Rh_{1}] on TiO_{2}(110)},
author = {Panukorn Sombut and Lena Puntscher and Marlene Atzmüller and Zdenek Jakub and Michele Reticcioli and Matthias Meier and Gareth S. Parkinson and Cesare Franchini},
doi = {10.1007/s11244-022-01651-0},
year = {2022},
date = {2022-07-25},
journal = {Topics in Catalysis},
volume = {65},
pages = {1620--1630},
abstract = {The local environment of metal-oxide supported single-atom catalysts plays a decisive role in the surface reactivity and related catalytic properties. The study of such systems is complicated by the presence of point defects on the surface, which are often associated with the localization of excess charge in the form of polarons. This can affect the stability, the electronic configuration, and the local geometry of the adsorbed adatoms. In this work, through the use of density functional theory and surface-sensitive experiments, we study the adsorption of Rh_{1}, Pt_{1}, and Au_{1} metals on the reduced TiO_{2}(110) surface, a prototypical polaronic material. A systematic analysis of the adsorption configurations and oxidation states of the adsorbed metals reveals different types of couplings between adsorbates and polarons. As confirmed by scanning tunneling microscopy measurements, the favored Pt_{1} and Au_{1} adsorption at oxygen vacancy sites is associated with a strong electronic charge transfer from polaronic states to adatom orbitals, which results in a reduction of the adsorbed metal. In contrast, the Rh_{1} adatoms interact weakly with the excess charge, which leaves the polarons largely unaffected. Our results show that an accurate understanding of the properties of single-atom catalysts on oxide surfaces requires a careful account of the interplay between adatoms, vacancy sites, and polarons.},
keywords = {P04, P07},
pubstate = {published},
tppubtype = {article}
}