Catalysis by ultrathin
LaBO3 (B=Co, Fe) perovskite films
Subproject P08
Perovskites are important catalysts, but detailed knowledge of their surface structure and chemistry is often lacking. The long-term objective of P08 is to elucidate structure-function correlations and visualize molecule-perovskite interaction in reactions involving O2, H2, CO, CO2, or H2O.
In the first project period, we will develop surface science-based model systems of LaCoO3 and LaFeO3 perovskites. Both epitaxial and polycrystalline thin films will be grown in UHV, guided by characterization via LEED, SXRD, SEM/EBSD, XPS/UPS/LEIS, IRAS, and TPD. Isotopically (18O or 13C) labeled adsorbates or films will reveal how oxygen and oxygen-containing molecules are activated. We will analyze the data in close collaboration with theoretical groups who simulate structure, stability, and infrared spectra (P03 Kresse).
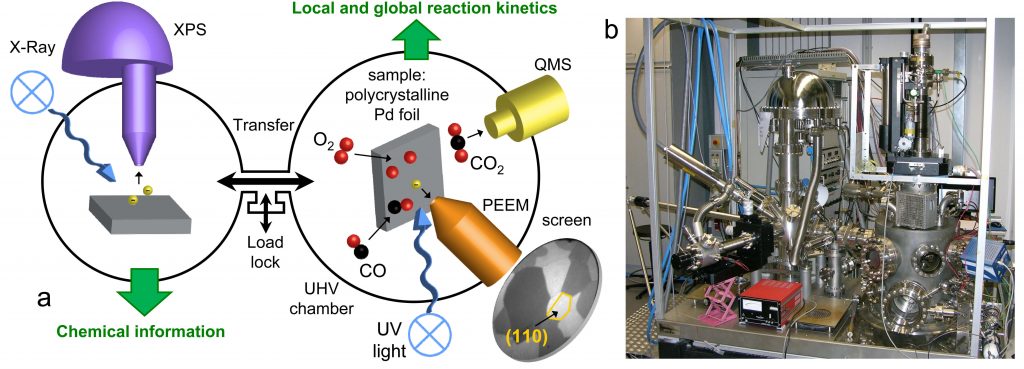
We will employ a unique combination of in situ surface spectroscopy (PM-IRAS, NAP-XPS, SXRD) and in situ surface microscopy (PEEM, SPEM), combined with MS gas phase analysis, to monitor ongoing reactions from HV to atmospheric pressure. This procedure should enable us to gain fundamental insights into the interplay of ternary oxide atomic and electronic structure, defects, composition, adsorption, as well as initiation and spatial progression of surface reactions on the mesoscale via reaction fronts (local kinetics by imaging). Project P08 will create the required bridge between single crystals (P02 Diebold, P04 Parkinson) and more application-relevant nanomaterials (P10 Föttinger).
Expertise
Our expertise is experimental surface science and its application to studies in heterogeneous catalysis. We operate a total of seven ultrahigh-vacuum (UHV) chambers, three of which are coupled to high-pressure cells. In situ and operando studies of surface reactions are carried out by area-averaging surface spectroscopy and real-time surface microscopy on the nano- and mesoscale. All chambers are equipped with facilities for sample preparation (sputtering, annealing, gas dosing), as well as various growth techniques (e-beam evaporators, Knudsen cells, sputter deposition). Analysis techniques used in our research include:
- Auger Electron Spectroscopy (AES)
- Field Emission Microscopy (FEM)
- Field Ion Microscopy (FIM)
- Gas Chromatography (GC)
- Low-Energy Electron Diffraction (LEED)
- Low-Energy Ion Scattering (LEIS)
- Mass Spectroscopy (MS)
- PhotoEmission Electron Microscopy (PEEM)
- Polarization Modulation Infrared Reflection-Absorption Spectroscopy (PM-IRAS)
- Sum Frequency Generation (SFG)
- Scanning PhotoElectron Microscopy (SPEM)
- Scanning Tunneling Microscopy (STM)
- Temperature-Programmed Desorption (TPD)
- Ultraviolet Photoelectron Spectroscopy (UPS)
- X-ray Absorption Spectroscopy (XAS)
- Surface X-Ray Diffraction (SXRD)
- X-ray Photoelectron Spectroscopy (XPS)
Collaboration Partners:
- Prof. Andreas Stierle, DESY Hamburg, Germany: SXRD
- Dr. Luca Gregoratti, ELETTRA Sincrotrone Trieste, Italy: SPEM
The combined application of photoemission electron microscopy (PEEM) and scanning photoelectron microscopy (SPEM) is particularly beneficial for TACO because these techniques allow visualizing ongoing reactions and local surface analysis on a µm-scale.
Team

Associates
Former Members
Publications
2023

Latschka, Markus; Wellscheid, Björn; Rameshan, Raffael; Schöberl, Tobias; Essmeister, Johannes; Pacholik, Gernot; Valentini, Francesco; Balta, Laura; Limbeck, Andreas; Kählig, Hanspeter; Föttinger, Karin
Influence of hot liquid flowing water on Zeolite Y stability
Journal ArticleOpen AccessIn: Microporous and Mesoporous Materials, vol. 354, no. 112557, 2023.
Abstract | Links | BibTeX | Tags: P10
@article{Latschka2023,
title = {Influence of hot liquid flowing water on Zeolite Y stability},
author = {Markus Latschka and Björn Wellscheid and Raffael Rameshan and Tobias Schöberl and Johannes Essmeister and Gernot Pacholik and Francesco Valentini and Laura Balta and Andreas Limbeck and Hanspeter Kählig and Karin Föttinger},
doi = {10.1016/j.micromeso.2023.112557},
year = {2023},
date = {2023-04-15},
urldate = {2023-04-15},
journal = {Microporous and Mesoporous Materials},
volume = {354},
number = {112557},
abstract = {Zeolite Y is used in a wide field of catalysis because of its high surface area and strong acidity. Since flowing water is present in many catalytic liquid phase reactions, its impact was investigated. For that, the zeolite Y was treated with water at 200 °C and 42 bar in a flow reactor. The resulting characterization showed strong structural changes at high water flows. The typical zeolite structure was almost completely lost, but an amorphous phase similar to the faujasite framework was formed. Due to this, the characteristic micropores were destroyed (d = 0.7 nm, volume was reduced from 0.18 to 0.01 cm³/g) and small mesopores were created (d = 2–3 nm, volume was increased from 0.25 to 0.51 cm³/g). As a result, the specific surface area was not greatly reduced and was still at around 250 m²/g. In addition, the amount of octahedrally coordinated EFAl increased from 54 to 70% and a γ-Al_{2}O_{3} as well as a kaolinite phase was observed. The formed tetrahedrally coordinated EFAl is responsible for EFAl-OH groups, which are strong Brønsted acid sites. In general, the total acid sites of the zeolite Y were not strongly reduced and the ratio of Lewis to Brønsted acid sites slightly increased from 70:30% to 80:20%. For all Al species, the oxygen coordination was strongly distorted. After water treatment, on Si a large number of coordinated OSi and OAl groups were substituted with OH groups. The ratio of Si to Al decreased from 1 to 0.7, because Si was dissolved out of the zeolite by the water. On the surface, it was vice versa, there the Si accumulated (the Si/Al ratio increased from 0.2 to 0.8), presumably as silica gel.},
keywords = {P10},
pubstate = {published},
tppubtype = {article}
}
2020
Pramhaas, Verena; Roiaz, Matteo; Bosio, Noemi; Corva, Manuel; Rameshan, Christoph; Vesselli, Erik; Grönbeck, Henrik; Rupprechter, Günther
Journal ArticleOpen AccessIn: ACS Catalysis, vol. 11, no. 1, pp. 208–214, 2020.
Abstract | Links | BibTeX | Tags: P08, P10, pre-TACO
@article{Pramhaas2020,
title = {Interplay between CO Disproportionation and Oxidation: On the Origin of the CO Reaction Onset on Atomic Layer Deposition-Grown Pt/ZrO_{2} Model Catalysts},
author = {Verena Pramhaas and Matteo Roiaz and Noemi Bosio and Manuel Corva and Christoph Rameshan and Erik Vesselli and Henrik Grönbeck and Günther Rupprechter},
doi = {10.1021/acscatal.0c03974},
year = {2020},
date = {2020-12-17},
urldate = {2020-12-17},
journal = {ACS Catalysis},
volume = {11},
number = {1},
pages = {208--214},
publisher = {American Chemical Society (ACS)},
abstract = {Pt/ZrO_{2} model catalysts were prepared by atomic layer deposition (ALD) and examined at mbar pressure by operando sum frequency generation (SFG) spectroscopy and near-ambient pressure X-ray photoelectron spectroscopy (NAP-XPS) combined with differentially pumped mass spectrometry (MS). ALD enables creating model systems ranging from Pt nanoparticles to bulk-like thin films. Polarization-dependent SFG of CO adsorption reveals both the adsorption configuration and the Pt particle morphology. By combining experimental data with ab initio density functional theory (DFT) calculations, we show that the CO reaction onset is determined by a delicate balance between CO disproportionation (Boudouard reaction) and oxidation. CO disproportionation occurs on low-coordinated Pt sites, but only at high CO coverages and when the remaining C atom is stabilized by a favorable coordination. Thus, under the current conditions, initial CO oxidation is found to be strongly influenced by the removal of carbon deposits formed through disproportionation mechanisms rather than being determined by the CO and oxygen inherent activity. Accordingly, at variance with the general expectation, rough Pt nanoparticles are seemingly less active than smoother Pt films. The applied approach enables bridging both the “materials and pressure gaps”.},
keywords = {P08, P10, pre-TACO},
pubstate = {published},
tppubtype = {article}
}

Haunold, Thomas; Rameshan, Christoph; Bukhtiyarov, Andrey V; Rupprechter, Günther
Journal ArticleOpen AccessIn: Review of Scientific Instruments, vol. 91, no. 12, pp. 125101, 2020.
Abstract | Links | BibTeX | Tags: P08, P10, pre-TACO
@article{Haunold2020,
title = {An ultrahigh vacuum-compatible reaction cell for model catalysis under atmospheric pressure flow conditions},
author = {Thomas Haunold and Christoph Rameshan and Andrey V Bukhtiyarov and Günther Rupprechter},
doi = {10.1063/5.0026171},
year = {2020},
date = {2020-12-01},
urldate = {2020-12-01},
journal = {Review of Scientific Instruments},
volume = {91},
number = {12},
pages = {125101},
publisher = {AIP Publishing},
abstract = {Atmospheric pressure reactions on model catalysts are typically performed in so-called high-pressure cells, with product analysis performed by gas chromatography (GC) or mass spectrometry (MS). However, in most cases, these cells have a large volume (liters) so that the reactions on catalysts with only cm^{2} surface area can be carried out only in the (recirculated) batch mode to accumulate sufficient product amounts. Herein, we describe a novel small-volume (milliliters) catalytic reactor that enables kinetic studies under atmospheric pressure flow conditions. The cell is located inside an ultrahigh vacuum chamber that is deliberately limited to basic functions. Model catalyst samples are mounted inside the reactor cell, which is locked to an oven for external heating and closed by using an extendable/retractable gas dosing tube. Reactant and product analyses are performed by both micro-GC and MS. The functionality of the new design is demonstrated by catalytic ethylene (C_{2}H_{4}) hydrogenation on polycrystalline Pt and Pd foils.},
keywords = {P08, P10, pre-TACO},
pubstate = {published},
tppubtype = {article}
}

Lindenthal, Lorenz; Rameshan, Raffael; Summerer, Harald; Ruh, Thomas; Popovic, Janko; Nenning, Andreas; Löffler, Stefan; Opitz, Alexander Karl; Blaha, Peter; Rameshan, Christoph
Modifying the Surface Structure of Perovskite-Based Catalysts by Nanoparticle Exsolution
Journal ArticleOpen AccessIn: Catalysts, vol. 10, no. 3, pp. 268, 2020.
Abstract | Links | BibTeX | Tags: P10, pre-TACO
@article{Lindenthal2020,
title = {Modifying the Surface Structure of Perovskite-Based Catalysts by Nanoparticle Exsolution},
author = {Lorenz Lindenthal and Raffael Rameshan and Harald Summerer and Thomas Ruh and Janko Popovic and Andreas Nenning and Stefan Löffler and Alexander Karl Opitz and Peter Blaha and Christoph Rameshan},
doi = {10.3390/catal10030268},
year = {2020},
date = {2020-03-01},
urldate = {2020-03-01},
journal = {Catalysts},
volume = {10},
number = {3},
pages = {268},
publisher = {MDPI AG},
abstract = {In heterogeneous catalysis, surfaces decorated with uniformly dispersed, catalytically-active (nano)particles are a key requirement for excellent performance. Beside standard catalyst preparation routines—with limitations in controlling catalyst surface structure (i.e., particle size distribution or dispersion)—we present here a novel time efficient route to precisely tailor catalyst surface morphology and composition of perovskites. Perovskite-type oxides of nominal composition ABO3 with transition metal cations on the B-site can exsolve the B-site transition metal upon controlled reduction. In this exsolution process, the transition metal emerges from the oxide lattice and migrates to the surface where it forms catalytically active nanoparticles. Doping the B-site with reducible and catalytically highly active elements, offers the opportunity of tailoring properties of exsolution catalysts. Here, we present the synthesis of two novel perovskite catalysts Nd_{0.6}Ca_{0.4}FeO_{3-δ} and Nd_{0.6}Ca_{0.4}Fe_{0.9}Co_{0.1}O_{3-δ} with characterisation by (in situ) XRD, SEM/TEM and XPS, supported by theory (DFT+U). Fe nanoparticle formation was observed for Nd_{0.6}Ca_{0.4}FeO_{3-δ}. In comparison, B site cobalt doping leads, already at lower reduction temperatures, to formation of finely dispersed Co nanoparticles on the surface. These novel perovskite-type catalysts are highly promising for applications in chemical energy conversion. First measurements revealed that exsolved Co nanoparticles significantly improve the catalytic activity for CO_{2} activation via reverse water gas shift reaction. },
keywords = {P10, pre-TACO},
pubstate = {published},
tppubtype = {article}
}
2018
Lukashuk, Liliana; Yigit, Nevzat; Rameshan, Raffael; Kolar, Elisabeth; Teschner, Detre; Hävecker, Michael; Knop-Gericke, Axel; Schlögl, Robert; Föttinger, Karin; Rupprechter, Günther
Operando Insights into CO Oxidation on Cobalt Oxide Catalysts by NAP-XPS, FTIR, and XRD
Journal ArticleOpen AccessIn: ACS Catalysis, vol. 8, no. 9, pp. 8630–8641, 2018.
Abstract | Links | BibTeX | Tags: P08, P10, pre-TACO
@article{Lukashuk2018,
title = {Operando Insights into CO Oxidation on Cobalt Oxide Catalysts by NAP-XPS, FTIR, and XRD},
author = {Liliana Lukashuk and Nevzat Yigit and Raffael Rameshan and Elisabeth Kolar and Detre Teschner and Michael Hävecker and Axel Knop-Gericke and Robert Schlögl and Karin Föttinger and Günther Rupprechter},
doi = {10.1021/acscatal.8b01237},
year = {2018},
date = {2018-08-07},
urldate = {2018-08-07},
journal = {ACS Catalysis},
volume = {8},
number = {9},
pages = {8630--8641},
publisher = {American Chemical Society (ACS)},
abstract = {Cobalt oxide Co_{3}O_{4} has recently emerged as promising, noble metal-free catalyst for oxidation reactions but a better understanding of the active catalyst under working conditions is required for further development and potential commercialization. An operando approach has been applied, combining near ambient (atmospheric) pressure X-ray photoelectron spectroscopy (NAP-XPS), Fourier transform infrared spectroscopy (FTIR), or X-ray diffraction (XRD) with simultaneous catalytic tests of CO oxidation on Co_{3}O_{4}, enabling one to monitor surface and bulk states under various reaction conditions (steady-state and dynamic conditions switching between CO and O_{2}). On the basis of the surface-specific chemical information a complex network of different reaction pathways unfolded: Mars-van-Krevelen (MvK), CO dissociation followed by carbon oxidation, and formation of carbonates. A possible Langmuir–Hinshelwood (LH) pathway cannot be excluded because of the good activity when no oxygen vacancies were detected. The combined NAP-XPS/FTIR results are in line with a MvK mechanism above 100 °C, involving the Co^{3+}/Co^{2+} redox couple and oxygen vacancy formation. Under steady state, the Co_{3}O_{4} surface appeared oxidized and the amount of reduced Co^{2+} species at/near the surface remained low up to 200 °C. Only in pure CO, about 15% of surface reduction were detected, suggesting that the active sites are a minority species. The operando spectroscopic studies also revealed additional reaction pathways: CO dissociation followed by carbon reoxidation and carbonate formation and its decomposition. However, due to their thermal stability in various atmospheres, the carbonates are rather spectators and also CO dissociation seems a minor route. This study thus highlights the benefits of combining operando surface sensitive techniques to gain insight into catalytically active surfaces.},
keywords = {P08, P10, pre-TACO},
pubstate = {published},
tppubtype = {article}
}
2016

Lukashuk, Liliana; Föttinger, Karin; Kolar, Elisabeth; Rameshan, Christoph; Teschner, Detre; Hävecker, Michael; Knop-Gericke, Axel; Yigit, Nevzat; Li, Hao; McDermott, Eamon; Stöger-Pollach, Michael; Rupprechter, Günther
Operando XAS and NAP-XPS studies of preferential CO oxidation on Co3O4 and CeO2-Co3O4 catalysts
Journal ArticleOpen AccessIn: Journal of Catalysis, vol. 344, pp. 1–15, 2016.
Abstract | Links | BibTeX | Tags: P08, P10, pre-TACO
@article{Lukashuk2016,
title = {Operando XAS and NAP-XPS studies of preferential CO oxidation on Co_{3}O_{4} and CeO_{2}-Co_{3}O_{4 }catalysts},
author = {Liliana Lukashuk and Karin Föttinger and Elisabeth Kolar and Christoph Rameshan and Detre Teschner and Michael Hävecker and Axel Knop-Gericke and Nevzat Yigit and Hao Li and Eamon McDermott and Michael Stöger-Pollach and Günther Rupprechter},
doi = {10.1016/j.jcat.2016.09.002},
year = {2016},
date = {2016-12-01},
urldate = {2016-12-01},
journal = {Journal of Catalysis},
volume = {344},
pages = {1--15},
publisher = {Elsevier BV},
abstract = {Co_{3}O_{4} is a promising catalyst for removing CO from H_{2} streams via the preferential CO oxidation (PROX). A Mars-van-Krevelen redox mechanism is often suggested but a detailed knowledge especially of the oxidation state of the catalytically active surface under reaction conditions is typically missing. We have thus utilized operando X-ray absorption spectroscopy to examine structure and oxidation state during PROX, and near atmospheric pressure-XPS at low photoelectron kinetic energies and thus high surface sensitivity to monitor surface composition changes. The rather easy surface reduction in pure CO (starting already at ∼100 °C) and the easy reoxidation by O_{2} suggest that molecularly adsorbed CO reacts with lattice oxygen, which is replenished by gas phase O_{2}. Nevertheless, the steady state concentration of oxygen vacancies under reaction conditions is too low even for XPS detection so that both the bulk and surface of Co_{3}O_{4} appear fully oxidized during PROX. Furthermore, the effect of adding CeO_{2} (a less active material) to Co_{3}O_{4} was studied. Promotion of Co_{3}O_{4} with 10 wt% CeO_{2} increases the reduction temperatures in CO and H_{2} and enhances the PROX activity. Since CeO_{2} is a less active material, this can only be explained by a higher activity of the Co-O-Ce interface.},
keywords = {P08, P10, pre-TACO},
pubstate = {published},
tppubtype = {article}
}

Wolfbeisser, Astrid; Sophiphun, Onsulang; Bernardi, Johannes; Wittayakun, Jatuporn; Föttinger, Karin; Rupprechter, Günther
Methane dry reforming over ceria-zirconia supported Ni catalysts
Journal ArticleOpen AccessIn: Catalysis Today, vol. 277, pp. 234–245, 2016.
Abstract | Links | BibTeX | Tags: P08, P10, pre-TACO
@article{Wolfbeisser2016,
title = {Methane dry reforming over ceria-zirconia supported Ni catalysts},
author = {Astrid Wolfbeisser and Onsulang Sophiphun and Johannes Bernardi and Jatuporn Wittayakun and Karin Föttinger and Günther Rupprechter},
doi = {10.1016/j.cattod.2016.04.025},
year = {2016},
date = {2016-11-15},
urldate = {2016-11-15},
journal = {Catalysis Today},
volume = {277},
pages = {234--245},
publisher = {Elsevier BV},
abstract = {Nickel nanoparticles supported on Ce_{1-x}Zr_{x}O_{2} mixed oxides prepared by different synthesis methods, as well as Ni-ZrO_{2} and Ni-CeO_{2}, were evaluated for their catalytic performance in methane dry reforming (MDR). MDR is an interesting model reaction to evaluate the reactivity and surface chemistry of mixed oxides. Textural and structural properties were studied by N_{2} adsorption and XRD. Mixed oxide preparation by co-precipitation resulted in catalysts with higher surface area than that of pure ZrO_{2} or CeO_{2}. XRD analysis showed the formation of different Ce_{1-x}Zr_{x}O_{2} solid solutions depending on using a surfactant or not. The catalyst prepared by surfactant assisted co-precipitation was not active for methane dry reforming most likely because of the encapsulation of Ni particles by ceria-zirconia particles, as revealed by TEM and H_{2} chemisorption. The catalytic activity of the catalyst prepared by co-precipitation without surfactant was comparable to Ni-ZrO_{2}. Clearly, catalyst activity strongly depends on preparation and on the resulting phase composition rather than on nominal composition. Compared to Ni-ZrO_{2} the ceria-zirconia supported Ni catalyst did not achieve higher activity or stability for methane dry reforming but, nevertheless, the formation of filamentous carbon was strongly reduced (100 times less carbonaceous species). Consequently, using ceria-zirconia as a support material decreases the risk of reactor tube blocking.},
keywords = {P08, P10, pre-TACO},
pubstate = {published},
tppubtype = {article}
}
2014

Föttinger, Karin; Rupprechter, Günther
Journal ArticleIn: Accounts of Chemical Research, vol. 47, no. 10, pp. 3071–3079, 2014.
Abstract | Links | BibTeX | Tags: P08, P10, pre-TACO
@article{Foettinger2014,
title = {In Situ Spectroscopy of Complex Surface Reactions on Supported Pd–Zn, Pd–Ga, and Pd(Pt)–Cu Nanoparticles},
author = {Karin Föttinger and Günther Rupprechter},
doi = {10.1021/ar500220v},
year = {2014},
date = {2014-09-23},
journal = {Accounts of Chemical Research},
volume = {47},
number = {10},
pages = {3071--3079},
publisher = {American Chemical Society (ACS)},
abstract = {It is well accepted that catalytically active surfaces frequently adapt to the reaction environment (gas composition, temperature) and that relevant “active phases” may only be created and observed during the ongoing reaction. Clearly, this requires the application of in situ spectroscopy to monitor catalysts at work. While changes in structure and composition may already occur for monometallic single crystal surfaces, such changes are typically more severe for oxide supported nanoparticles, in particular when they are composed of two metals. The metals may form ordered intermetallic compounds (e.g. PdZn on ZnO, Pd_{2}Ga on Ga_{2}O_{3}) or disordered substitutional alloys (e.g. PdCu, PtCu on hydrotalcite). We discuss the formation and stability of bimetallic nanoparticles, focusing on the effect of atomic and electronic structure on catalytic selectivity for methanol steam reforming (MSR) and hydrodechlorination of trichloroethylene. Emphasis is placed on the in situ characterization of functioning catalysts, mainly by (polarization modulated) infrared spectroscopy, ambient pressure X-ray photoelectron spectroscopy, X-ray absorption near edge structure, and X-ray diffraction. In the present contribution, we pursue a two-fold, fundamental and applied, approach investigating technologically applied catalysts as well as model catalysts, which provides comprehensive and complementary information of the relevant surface processes at the atomic or molecular level. Comparison to results of theoretical simulations yields further insight.
Several key aspects were identified that control the nanoparticle functionality: (i) alloying (IMC formation) leads to site isolation of specific (e.g. Pd) atoms but also yields very specific electronic structure due to the (e.g. Zn or Ga or Cu) neighboring atoms; (i) for intermetallic PdZn, the thickness of the surface alloy, and its resulting valence band structure and corrugation, turned out to be critical for MSR selectivity; (ii) the limited stability of phases, such as Pd_{2}Ga under MSR conditions, also limits selectivity; (iii) favorably bimetallic catalysts act bifunctional, such as activating methanol AND water or decomposing trichlorothylene AND activating hydrogen; (iv) bifunctionality is achieved either by the two metals or by one metal and the metal–oxide interface; (v) intimate contact between the two interacting sites is required (that cannot be realized by two monometallic nanoparticles being just located close by).
The current studies illustrate how rather simple bimetallic nanoparticles may exhibit intriguing diversity and flexibility, exceeding by far the properties of the individual metals. It is also demonstrated how complex reactions can be elucidated with the help of in situ spectroscopy, in particular when complementary methods with varying surface sensitivity are applied.},
keywords = {P08, P10, pre-TACO},
pubstate = {published},
tppubtype = {article}
}
Several key aspects were identified that control the nanoparticle functionality: (i) alloying (IMC formation) leads to site isolation of specific (e.g. Pd) atoms but also yields very specific electronic structure due to the (e.g. Zn or Ga or Cu) neighboring atoms; (i) for intermetallic PdZn, the thickness of the surface alloy, and its resulting valence band structure and corrugation, turned out to be critical for MSR selectivity; (ii) the limited stability of phases, such as Pd2Ga under MSR conditions, also limits selectivity; (iii) favorably bimetallic catalysts act bifunctional, such as activating methanol AND water or decomposing trichlorothylene AND activating hydrogen; (iv) bifunctionality is achieved either by the two metals or by one metal and the metal–oxide interface; (v) intimate contact between the two interacting sites is required (that cannot be realized by two monometallic nanoparticles being just located close by).
The current studies illustrate how rather simple bimetallic nanoparticles may exhibit intriguing diversity and flexibility, exceeding by far the properties of the individual metals. It is also demonstrated how complex reactions can be elucidated with the help of in situ spectroscopy, in particular when complementary methods with varying surface sensitivity are applied.