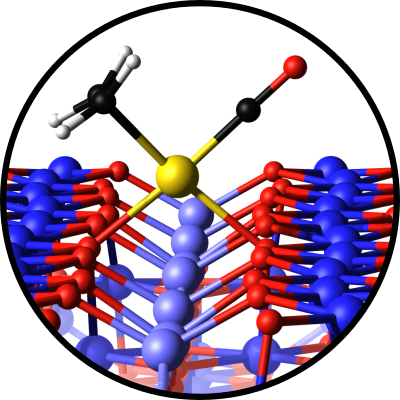
Atomic-scale studies of catalysis
by spinel oxides
Subproject P04
The spinel class of metal oxides hosts diverse materials, some of which make excellent catalysts. Fe3O4 is already the industrial catalyst for the high-temperature water-gas shift reaction (CO+H2O -> H2+CO2), but research is needed to identify the optimal replacement for the toxic Cr promoter/stabilizer. Ternary MeFe2O4 compounds (Me=Fe, Ni, Co, Mn) are active and stable for the electrochemical oxygen evolution reaction (OER). However, the structure of the active catalyst and the reaction mechanisms are unknown. While these reactions appear different, both clearly benefit from a combination of multivalent cations in the surface layers.
In this project, we will seek to learn why, using a combination of atomic-scale imaging, a host of spectroscopies, and theory. We will dope the Fe3O4(001) surface with 3d transition metals and investigate how the adsorption energies, XPS binding energies, and IRAS frequencies of H2O, CO, CO2, O2, and H2 change with sample composition all the way from isolated dopants to ternary thin films. We will use the data obtained in tightly-controlled UHV experiments to:
i) Interpret the reactivity of our model catalysts under realistic HTWGS and OER conditions.
ii) Provide the benchmark data for experiments on nominally similar powder catalysts (P10 Föttinger).
iii) Support the development of theoretical modeling (P07 Franchini).
A joint postdoc (P04-P11) will facilitate the new collaboration with P11 Backus.
Expertise
- Scanning Tunneling Microscopy (STM) (in UHV 4K – 300 K, electrochemical STM)
- Atomic Force Microscopy (AFM): UHV-based (q+ sensor) and in the ambient (cantilever-based)
- Low-Energy Electron Diffraction (LEED)
- Reflection High Energy Diffraction (RHEED)
- X-ray Photoelectron Spectroscopy (XPS)
- Ultraviolet Photoelectron Spectroscopy (UPS)
- Auger Electron Spectroscopy (AES)
- Low-energy He+ ion scattering (LEIS)
- Thermal Programmed Desorption Spectroscopy (TPD)
Team

Associates
Publications
2022
Meier, Matthias; Hulva, Jan; Jakub, Zdenek; Kraushofer, Florian; Bobić, Mislav; Bliem, Roland; Setvin, Martin; Schmid, Michael; Diebold, Ulrike; Franchini, Cesare; Parkinson, Gareth S.
Journal ArticleOpen AccessIn: ScienceAdvances, vol. 8, iss. 13, pp. eabn4580, 2022.
Abstract | Links | BibTeX | Tags: P02, P04, P07
@article{SCIADV2022,
title = {CO oxidation by Pt_{2}/Fe_{3}O_{4}: Metastable dimer and support configurations facilitate lattice oxygen extraction},
author = {Matthias Meier and Jan Hulva and Zdenek Jakub and Florian Kraushofer and Mislav Bobić and Roland Bliem and Martin Setvin and Michael Schmid and Ulrike Diebold and Cesare Franchini and Gareth S. Parkinson},
url = {https://www.science.org/doi/10.1126/sciadv.abn4580},
doi = {10.1126/sciadv.abn4580},
year = {2022},
date = {2022-04-01},
urldate = {2022-04-01},
journal = {ScienceAdvances},
volume = {8},
issue = {13},
pages = {eabn4580},
abstract = {Heterogeneous catalysts based on subnanometer metal clusters often exhibit strongly size-dependent properties, and the addition or removal of a single atom can make all the difference. Identifying the most active species and deciphering the reaction mechanism is extremely difficult, however, because it is often not clear how the catalyst evolves in operando. Here, we use a combination of atomically resolved scanning probe microscopies, spectroscopic techniques, and density functional theory (DFT)–based calculations to study CO oxidation by a model Pt/Fe_{3}O_{4}(001) “single-atom” catalyst. We demonstrate that (PtCO)_{2} dimers, formed dynamically through the agglomeration of mobile Pt-carbonyl species, catalyze a reaction involving the oxide support to form CO_{2}. Pt_{2} dimers produce one CO_{2} molecule before falling apart into two adatoms, releasing the second CO. Olattice extraction only becomes facile when both the Pt-dimer and the Fe_{3}O_{4} support can access metastable configurations, suggesting that substantial, concerted rearrangements of both cluster and support must be considered for reactions occurring at elevated temperature.},
keywords = {P02, P04, P07},
pubstate = {published},
tppubtype = {article}
}
2021
Jakub, Zdenek; Meier, Matthias; Kraushofer, Florian; Balajka, Jan; Pavelec, Jiri; Schmid, Michael; Franchini, Cesare; Diebold, Ulrike; Parkinson, Gareth S.
Rapid oxygen exchange between hematite and water vapor
Journal ArticleOpen AccessIn: Nature Communications, vol. 12, iss. 1, no. 6488, 2021.
Abstract | Links | BibTeX | Tags: P02, P04, P07
@article{Jakub2021,
title = {Rapid oxygen exchange between hematite and water vapor},
author = {Zdenek Jakub and Matthias Meier and Florian Kraushofer and Jan Balajka and Jiri Pavelec and Michael Schmid and Cesare Franchini and Ulrike Diebold and Gareth S. Parkinson},
doi = {10.1038/s41467-021-26601-4},
year = {2021},
date = {2021-11-10},
journal = {Nature Communications},
volume = {12},
number = {6488},
issue = {1},
publisher = {Springer Science and Business Media LLC},
abstract = {Oxygen exchange at oxide/liquid and oxide/gas interfaces is important in technology and environmental studies, as it is closely linked to both catalytic activity and material degradation. The atomic-scale details are mostly unknown, however, and are often ascribed to poorly defined defects in the crystal lattice. Here we show that even thermodynamically stable, well-ordered surfaces can be surprisingly reactive. Specifically, we show that all the 3-fold coordinated lattice oxygen atoms on a defect-free single-crystalline “r-cut” (1-102) surface of hematite (α-Fe_{2}O_{3}) are exchanged with oxygen from surrounding water vapor within minutes at temperatures below 70 °C, while the atomic-scale surface structure is unperturbed by the process. A similar behavior is observed after liquid-water exposure, but the experimental data clearly show most of the exchange happens during desorption of the final monolayer, not during immersion. Density functional theory computations show that the exchange can happen during on-surface diffusion, where the cost of the lattice oxygen extraction is compensated by the stability of an HO-HOH-OH complex. Such insights into lattice oxygen stability are highly relevant for many research fields ranging from catalysis and hydrogen production to geochemistry and paleoclimatology.},
keywords = {P02, P04, P07},
pubstate = {published},
tppubtype = {article}
}
Mirabella, Francesca; Müllner, Matthias; Touzalin, Thomas; Riva, Michele; Jakub, Zdenek; Kraushofer, Florian; Schmid, Michael; Koper, Marc T M; Parkinson, Gareth S.; Diebold, Ulrike
Journal ArticleOpen AccessIn: Electrochimica Acta, vol. 389, pp. 138638, 2021.
Abstract | Links | BibTeX | Tags: P02, P04, pre-TACO
@article{Mirabella2021,
title = {Ni-modified Fe_{3}O_{4}(001) surface as a simple model system for understanding the oxygen evolution reaction},
author = {Francesca Mirabella and Matthias Müllner and Thomas Touzalin and Michele Riva and Zdenek Jakub and Florian Kraushofer and Michael Schmid and Marc T M Koper and Gareth S. Parkinson and Ulrike Diebold},
doi = {10.1016/j.electacta.2021.138638},
year = {2021},
date = {2021-09-01},
urldate = {2021-09-01},
journal = {Electrochimica Acta},
volume = {389},
pages = {138638},
publisher = {Elsevier BV},
abstract = {Electrochemical water splitting is an environmentally friendly technology to store renewable energy in the form of chemical fuels. Among the earth-abundant first-row transition metal-based catalysts, mixed Ni-Fe oxides have shown promising performance for effective and low-cost catalysis of the oxygen evolution reaction (OER) in alkaline media, but the synergistic roles of Fe and Ni cations in the OER mechanism remain unclear. In this work, we report how addition of Ni changes the reactivity of a model iron oxide catalyst, based on Ni deposited on and incorporated in a magnetite Fe_{3}O_{4}(001) single crystal, using a combination of surface science techniques in ultra-high vacuum such as low energy electron diffraction (LEED), x-ray photoelectron spectroscopy (XPS), low-energy ion scattering (LEIS), and scanning tunneling microscopy (STM), as well as atomic force microscopy (AFM) in air, and electrochemical methods such as cyclic voltammetry (CV) and electrochemical impedance spectroscopy (EIS) in alkaline media. A significant improvement in the OER activity is observed when the top surface presents an iron fraction among the cations in the range of 20-40%, which is in good agreement with what has been observed for powder catalysts. Furthermore, a decrease in the OER overpotential is observed following surface aging in electrolyte for three days. At higher Ni load, AFM shows the growth of a new phase attributed to an (oxy)-hydroxide phase which, according to CV measurements, does not seem to correlate with the surface activity towards OER. EIS suggests that the OER precursor species observed on the clean and Ni-modified surfaces are similar and Fe-centered, but form at lower overpotentials when the surface Fe:Ni ratio is optimized. We propose that the well-defined Fe_{3}O_{4}(001) surface can serve as a model system for understanding the OER mechanism and establishing the structure-reactivity relation on mixed Fe-Ni oxides.},
keywords = {P02, P04, pre-TACO},
pubstate = {published},
tppubtype = {article}
}

Hulva, Jan; Meier, Matthias; Bliem, Roland; Jakub, Zdenek; Kraushofer, Florian; Schmid, Michael; Diebold, Ulrike; Franchini, Cesare; Parkinson, Gareth S.
Unraveling CO adsorption on model single-atom catalysts
Journal ArticleIn: Science, vol. 371, no. 6527, pp. 375–379, 2021.
Abstract | Links | BibTeX | Tags: P02, P04, P07, pre-TACO
@article{Hulva2021,
title = {Unraveling CO adsorption on model single-atom catalysts},
author = {Jan Hulva and Matthias Meier and Roland Bliem and Zdenek Jakub and Florian Kraushofer and Michael Schmid and Ulrike Diebold and Cesare Franchini and Gareth S. Parkinson},
doi = {10.1126/science.abe5757},
year = {2021},
date = {2021-01-22},
urldate = {2021-01-22},
journal = {Science},
volume = {371},
number = {6527},
pages = {375--379},
publisher = {American Association for the Advancement of Science (AAAS)},
abstract = {Understanding how the local environment of a “single-atom” catalyst affects stability and reactivity remains a challenge. We present an in-depth study of copper_{1}, silver_{1}, gold_{1}, nickel_{1}, palladium_{1}, platinum_{1}, rhodium_{1}, and iridium_{1} species on Fe_{3}O_{4}(001), a model support in which all metals occupy the same twofold-coordinated adsorption site upon deposition at room temperature. Surface science techniques revealed that CO adsorption strength at single metal sites differs from the respective metal surfaces and supported clusters. Charge transfer into the support modifies the d-states of the metal atom and the strength of the metal–CO bond. These effects could strengthen the bond (as for Ag_{1}–CO) or weaken it (as for Ni_{1}–CO), but CO-induced structural distortions reduce adsorption energies from those expected on the basis of electronic structure alone. The extent of the relaxations depends on the local geometry and could be predicted by analogy to coordination chemistry.},
keywords = {P02, P04, P07, pre-TACO},
pubstate = {published},
tppubtype = {article}
}
2020

Timmermann, Jakob; Kraushofer, Florian; Resch, Nikolaus; Li, Peigang; Wang, Yu; Mao, Zhiqiang; Riva, Michele; Lee, Yonghyuk; Staacke, Carsten; Schmid, Michael; Scheurer, Christoph; Parkinson, Gareth S.; Diebold, Ulrike; Reuter, Karsten
IrO2 Surface Complexions Identified through Machine Learning and Surface Investigations
Journal ArticleIn: Physical Review Letters, vol. 125, no. 20, pp. 206101, 2020.
Abstract | Links | BibTeX | Tags: P02, P04, pre-TACO
@article{Timmermann2020,
title = {IrO_{2} Surface Complexions Identified through Machine Learning and Surface Investigations},
author = {Jakob Timmermann and Florian Kraushofer and Nikolaus Resch and Peigang Li and Yu Wang and Zhiqiang Mao and Michele Riva and Yonghyuk Lee and Carsten Staacke and Michael Schmid and Christoph Scheurer and Gareth S. Parkinson and Ulrike Diebold and Karsten Reuter},
doi = {10.1103/physrevlett.125.206101},
year = {2020},
date = {2020-11-10},
urldate = {2020-11-10},
journal = {Physical Review Letters},
volume = {125},
number = {20},
pages = {206101},
publisher = {American Physical Society (APS)},
abstract = {A Gaussian approximation potential was trained using density-functional theory data to enable a global geometry optimization of low-index rutile IrO_{2} facets through simulated annealing. Ab initio thermodynamics identifies (101) and (111) (1×1) terminations competitive with (110) in reducing environments. Experiments on single crystals find that (101) facets dominate and exhibit the theoretically predicted (1×1) periodicity and x-ray photoelectron spectroscopy core-level shifts. The obtained structures are analogous to the complexions discussed in the context of ceramic battery materials.},
keywords = {P02, P04, pre-TACO},
pubstate = {published},
tppubtype = {article}
}

Grumelli, Doris; Wiegmann, Tim; Barja, Sara; Reikowski, Finn; Maroun, Fouad; Allongue, Philippe; Balajka, Jan; Parkinson, Gareth S.; Diebold, Ulrike; Kern, Klaus; Magnussen, Olaf M
Electrochemical Stability of the Reconstructed Fe3O4(001) Surface
Journal ArticleIn: Angewandte Chemie - International Edition, vol. 59, no. 49, pp. 21904–21908, 2020.
Abstract | Links | BibTeX | Tags: P02, P04, pre-TACO
@article{Grumelli2020,
title = {Electrochemical Stability of the Reconstructed Fe_{3}O_{4}(001) Surface},
author = {Doris Grumelli and Tim Wiegmann and Sara Barja and Finn Reikowski and Fouad Maroun and Philippe Allongue and Jan Balajka and Gareth S. Parkinson and Ulrike Diebold and Klaus Kern and Olaf M Magnussen},
doi = {10.1002/anie.202008785},
year = {2020},
date = {2020-07-29},
urldate = {2020-07-29},
journal = {Angewandte Chemie - International Edition},
volume = {59},
number = {49},
pages = {21904--21908},
publisher = {Wiley},
abstract = {Establishing the atomic-scale structure of metal-oxide surfaces during electrochemical reactions is a key step to modeling this important class of electrocatalysts. Here, we demonstrate that the characteristic (√2×√2)R45° surface reconstruction formed on (001)-oriented magnetite single crystals is maintained after immersion in 0.1 M NaOH at 0.20 V vs. Ag/AgCl and we investigate its dependence on the electrode potential. We follow the evolution of the surface using in situ and operando surface X-ray diffraction from the onset of hydrogen evolution, to potentials deep in the oxygen evolution reaction (OER) regime. The reconstruction remains stable for hours between −0.20 and 0.60 V and, surprisingly, is still present at anodic current densities of up to 10 mA cm^{−2} and strongly affects the OER kinetics. We attribute this to a stabilization of the Fe_{3}O_{4} bulk by the reconstructed surface. At more negative potentials, a gradual and largely irreversible lifting of the reconstruction is observed due to the onset of oxide reduction.},
keywords = {P02, P04, pre-TACO},
pubstate = {published},
tppubtype = {article}
}
2019

Jakub, Zdenek; Hulva, Jan; Meier, Matthias; Bliem, Roland; Kraushofer, Florian; Setvin, Martin; Schmid, Michael; Diebold, Ulrike; Franchini, Cesare; Parkinson, Gareth S.
Local Structure and Coordination Define Adsorption in a Model Ir1/Fe3O4 Single-Atom Catalyst
Journal ArticleOpen AccessIn: Angewandte Chemie - International Edition, vol. 58, no. 39, pp. 13961–13968, 2019.
Abstract | Links | BibTeX | Tags: P02, P04, P07, pre-TACO
@article{Jakub2019,
title = {Local Structure and Coordination Define Adsorption in a Model Ir_{1}/Fe_{3}O_{4} Single-Atom Catalyst},
author = {Zdenek Jakub and Jan Hulva and Matthias Meier and Roland Bliem and Florian Kraushofer and Martin Setvin and Michael Schmid and Ulrike Diebold and Cesare Franchini and Gareth S. Parkinson},
doi = {10.1002/anie.201907536},
year = {2019},
date = {2019-07-24},
urldate = {2019-07-24},
journal = {Angewandte Chemie - International Edition},
volume = {58},
number = {39},
pages = {13961--13968},
publisher = {Wiley},
abstract = {Single-atom catalysts (SACs) bridge homo- and heterogeneous catalysis because the active site is a metal atom coordinated to surface ligands. The local binding environment of the atom should thus strongly influence how reactants adsorb. Now, atomically resolved scanning-probe microscopy, X-ray photoelectron spectroscopy, temperature-programmed desorption, and DFT are used to study how CO binds at different Ir_{1} sites on a precisely defined Fe_{3}O_{4}(001) support. The two- and five-fold-coordinated Ir adatoms bind CO more strongly than metallic Ir, and adopt structures consistent with square-planar Ir^{I} and octahedral Ir^{III} complexes, respectively. Ir incorporates into the subsurface already at 450 K, becoming inactive for adsorption. Above 900 K, the Ir adatoms agglomerate to form nanoparticles encapsulated by iron oxide. These results demonstrate the link between SAC systems and coordination complexes, and that incorporation into the support is an important deactivation mechanism.},
keywords = {P02, P04, P07, pre-TACO},
pubstate = {published},
tppubtype = {article}
}