Publications
2020
Lesnicki, Dominika; Zhang, Zhen; Bonn, Mischa; Sulpizi, Marialore; Backus, Ellen H. G.
Journal ArticleOpen AccessIn: Angewandte Chemie - International Edition, vol. 59, no. 31, pp. 13116–13121, 2020.
Abstract | Links | BibTeX | Tags: P11, pre-TACO
@article{Lesnicki2020,
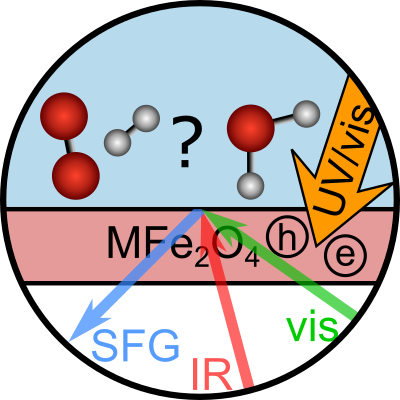
title = {Surface Charges at the CaF_{2}/Water Interface Allow Very Fast Intermolecular Vibrational-Energy Transfer},
author = {Dominika Lesnicki and Zhen Zhang and Mischa Bonn and Marialore Sulpizi and Ellen H. G. Backus},
doi = {10.1002/anie.202004686},
year = {2020},
date = {2020-04-02},
urldate = {2020-04-02},
journal = {Angewandte Chemie - International Edition},
volume = {59},
number = {31},
pages = {13116--13121},
publisher = {Wiley},
abstract = {We investigate the dynamics of water in contact with solid calcium fluoride, where at low pH, localized charges can develop upon fluorite dissolution. We use 2D surface-specific vibrational spectroscopy to quantify the heterogeneity of the interfacial water (D_{2}O) molecules and provide information about the sub-picosecond vibrational-energy-relaxation dynamics at the buried solid/liquid interface. We find that strongly H-bonded OD groups, with a vibrational frequency below 2500 cm^{−1}, display very rapid spectral diffusion and vibrational relaxation; for weakly H-bonded OD groups, above 2500 cm^{−1}, the dynamics slows down substantially. Atomistic simulations based on electronic-structure theory reveal the molecular origin of energy transport through the local H-bond network. We conclude that strongly oriented H-bonded water molecules in the adsorbed layer, whose orientation is pinned by the localized charge defects, can exchange vibrational energy very rapidly due to the strong collective dipole, compensating for a partially missing solvation shell.},
keywords = {P11, pre-TACO},
pubstate = {published},
tppubtype = {article}
}

Lindenthal, Lorenz; Rameshan, Raffael; Summerer, Harald; Ruh, Thomas; Popovic, Janko; Nenning, Andreas; Löffler, Stefan; Opitz, Alexander Karl; Blaha, Peter; Rameshan, Christoph
Modifying the Surface Structure of Perovskite-Based Catalysts by Nanoparticle Exsolution
Journal ArticleOpen AccessIn: Catalysts, vol. 10, no. 3, pp. 268, 2020.
Abstract | Links | BibTeX | Tags: P10, pre-TACO
@article{Lindenthal2020,
title = {Modifying the Surface Structure of Perovskite-Based Catalysts by Nanoparticle Exsolution},
author = {Lorenz Lindenthal and Raffael Rameshan and Harald Summerer and Thomas Ruh and Janko Popovic and Andreas Nenning and Stefan Löffler and Alexander Karl Opitz and Peter Blaha and Christoph Rameshan},
doi = {10.3390/catal10030268},
year = {2020},
date = {2020-03-01},
urldate = {2020-03-01},
journal = {Catalysts},
volume = {10},
number = {3},
pages = {268},
publisher = {MDPI AG},
abstract = {In heterogeneous catalysis, surfaces decorated with uniformly dispersed, catalytically-active (nano)particles are a key requirement for excellent performance. Beside standard catalyst preparation routines—with limitations in controlling catalyst surface structure (i.e., particle size distribution or dispersion)—we present here a novel time efficient route to precisely tailor catalyst surface morphology and composition of perovskites. Perovskite-type oxides of nominal composition ABO3 with transition metal cations on the B-site can exsolve the B-site transition metal upon controlled reduction. In this exsolution process, the transition metal emerges from the oxide lattice and migrates to the surface where it forms catalytically active nanoparticles. Doping the B-site with reducible and catalytically highly active elements, offers the opportunity of tailoring properties of exsolution catalysts. Here, we present the synthesis of two novel perovskite catalysts Nd_{0.6}Ca_{0.4}FeO_{3-δ} and Nd_{0.6}Ca_{0.4}Fe_{0.9}Co_{0.1}O_{3-δ} with characterisation by (in situ) XRD, SEM/TEM and XPS, supported by theory (DFT+U). Fe nanoparticle formation was observed for Nd_{0.6}Ca_{0.4}FeO_{3-δ}. In comparison, B site cobalt doping leads, already at lower reduction temperatures, to formation of finely dispersed Co nanoparticles on the surface. These novel perovskite-type catalysts are highly promising for applications in chemical energy conversion. First measurements revealed that exsolved Co nanoparticles significantly improve the catalytic activity for CO_{2} activation via reverse water gas shift reaction. },
keywords = {P10, pre-TACO},
pubstate = {published},
tppubtype = {article}
}
2019
Michl, Jakob; Sega, Marcello; Dellago, Christoph
Phase stability of the ice XVII-based CO2 chiral hydrate from molecular dynamics simulations
Journal ArticleOpen AccessIn: The Journal of Chemical Physics, vol. 151, no. 10, pp. 104502, 2019.
Abstract | Links | BibTeX | Tags: P12, pre-TACO
@article{Michl2019,
title = {Phase stability of the ice XVII-based CO_{2} chiral hydrate from molecular dynamics simulations},
author = {Jakob Michl and Marcello Sega and Christoph Dellago},
doi = {10.1063/1.5116540},
year = {2019},
date = {2019-09-12},
urldate = {2019-09-12},
journal = {The Journal of Chemical Physics},
volume = {151},
number = {10},
pages = {104502},
publisher = {AIP Publishing},
abstract = {We computed the phase diagram of CO_{2} hydrates at high pressure (HP), from 0.3 to 20 kbar, by means of molecular dynamics simulations. The two CO_{2} hydrates known to occur in this pressure range are the cubic structure I (sI) clathrate and the HP hydrate, whose water framework is the recently discovered ice XVII. We investigated the stability of both hydrates upon heating (melting) as well as the phase changes upon compression. The CO_{2}-filled ice XVII is found to be more stable than the sI clathrate and than the mixture of ice VI and dry ice at pressure values ranging from 6 to 18 kbar and in a wide temperature range, although a phenomenological correction suggests that the stability should more realistically range from 6.5 to 13.5 kbar. Our simulation results support the current hypothesis that the HP hydrate is stable at temperatures above the melting curve of ice VI.},
keywords = {P12, pre-TACO},
pubstate = {published},
tppubtype = {article}
}
Jakub, Zdenek; Hulva, Jan; Meier, Matthias; Bliem, Roland; Kraushofer, Florian; Setvin, Martin; Schmid, Michael; Diebold, Ulrike; Franchini, Cesare; Parkinson, Gareth S.
Local Structure and Coordination Define Adsorption in a Model Ir1/Fe3O4 Single-Atom Catalyst
Journal ArticleOpen AccessIn: Angewandte Chemie - International Edition, vol. 58, no. 39, pp. 13961–13968, 2019.
Abstract | Links | BibTeX | Tags: P02, P04, P07, pre-TACO
@article{Jakub2019,
title = {Local Structure and Coordination Define Adsorption in a Model Ir_{1}/Fe_{3}O_{4} Single-Atom Catalyst},
author = {Zdenek Jakub and Jan Hulva and Matthias Meier and Roland Bliem and Florian Kraushofer and Martin Setvin and Michael Schmid and Ulrike Diebold and Cesare Franchini and Gareth S. Parkinson},
doi = {10.1002/anie.201907536},
year = {2019},
date = {2019-07-24},
urldate = {2019-07-24},
journal = {Angewandte Chemie - International Edition},
volume = {58},
number = {39},
pages = {13961--13968},
publisher = {Wiley},
abstract = {Single-atom catalysts (SACs) bridge homo- and heterogeneous catalysis because the active site is a metal atom coordinated to surface ligands. The local binding environment of the atom should thus strongly influence how reactants adsorb. Now, atomically resolved scanning-probe microscopy, X-ray photoelectron spectroscopy, temperature-programmed desorption, and DFT are used to study how CO binds at different Ir_{1} sites on a precisely defined Fe_{3}O_{4}(001) support. The two- and five-fold-coordinated Ir adatoms bind CO more strongly than metallic Ir, and adopt structures consistent with square-planar Ir^{I} and octahedral Ir^{III} complexes, respectively. Ir incorporates into the subsurface already at 450 K, becoming inactive for adsorption. Above 900 K, the Ir adatoms agglomerate to form nanoparticles encapsulated by iron oxide. These results demonstrate the link between SAC systems and coordination complexes, and that incorporation into the support is an important deactivation mechanism.},
keywords = {P02, P04, P07, pre-TACO},
pubstate = {published},
tppubtype = {article}
}

Singraber, Andreas; Morawietz, Tobias; Behler, Jörg; Dellago, Christoph
Parallel Multistream Training of High-Dimensional Neural Network Potentials
Journal ArticleIn: Journal of Chemical Theory and Computation, vol. 15, no. 5, pp. 3075–3092, 2019.
Abstract | Links | BibTeX | Tags: P12, pre-TACO
@article{Singraber2019,
title = {Parallel Multistream Training of High-Dimensional Neural Network Potentials},
author = {Andreas Singraber and Tobias Morawietz and Jörg Behler and Christoph Dellago},
doi = {10.1021/acs.jctc.8b01092},
year = {2019},
date = {2019-04-17},
journal = {Journal of Chemical Theory and Computation},
volume = {15},
number = {5},
pages = {3075--3092},
publisher = {American Chemical Society (ACS)},
abstract = {Over the past years high-dimensional neural network potentials (HDNNPs), fitted to accurately reproduce ab initio potential energy surfaces, have become a powerful tool in chemistry, physics and materials science. Here, we focus on the training of the neural networks that lies at the heart of the HDNNP method. We present an efficient approach for optimizing the weight parameters of the neural network via multistream Kalman filtering, using potential energies and forces as reference data. In this procedure, the choice of the free parameters of the Kalman filter can have a significant impact on the fit quality. Carrying out a large parameter study, we determine optimal settings and demonstrate how to optimize training results of HDNNPs. Moreover, we illustrate our HDNNP training approach by revisiting previously presented fits for water and developing a new potential for copper sulfide. This material, accessible in computer simulations so far only via first-principles methods, forms a particularly complex solid structure at low temperatures and undergoes a phase transition to a superionic state upon heating. Analyzing MD simulations carried out with the Cu_{2}S HDNNP, we confirm that the underlying ab initio reference method indeed reproduces this behavior.},
keywords = {P12, pre-TACO},
pubstate = {published},
tppubtype = {article}
}

Schlegel, Simon J; Hosseinpour, Saman; Gebhard, Maximilian; Devi, Anjana; Bonn, Mischa; Backus, Ellen H. G.
How water flips at charged titanium dioxide: an SFG-study on the water–TiO2 interface
Journal ArticleOpen AccessIn: Physical Chemistry Chemical Physics, vol. 21, no. 17, pp. 8956–8964, 2019.
Abstract | Links | BibTeX | Tags: P11, pre-TACO
@article{Schlegel2019,
title = {How water flips at charged titanium dioxide: an SFG-study on the water–TiO_{2} interface},
author = {Simon J Schlegel and Saman Hosseinpour and Maximilian Gebhard and Anjana Devi and Mischa Bonn and Ellen H. G. Backus},
doi = {10.1039/c9cp01131e},
year = {2019},
date = {2019-04-05},
urldate = {2019-04-05},
journal = {Physical Chemistry Chemical Physics},
volume = {21},
number = {17},
pages = {8956--8964},
publisher = {Royal Society of Chemistry (RSC)},
abstract = {Photocatalytic splitting of water into hydrogen and oxygen by utilizing sunlight and a photocatalyst is a promising way of generating clean energy. Here, we report a molecular-level study on heavy water (D_{2}O) interacting with TiO_{2} as a model photocatalyst. We employed the surface specific technique Sum-Frequency-Generation (SFG) spectroscopy to determine the nature of the hydrogen bonding environment and the orientation of interfacial water molecules using their OD-stretch vibrations as reporters. By examining solutions with various pD-values, we observe an intensity-minimum at around pD 5, corresponding to the balance of protonation and deprotonation of TiO_{2} (point of zero charge). The majority of water molecules’ deuterium atoms point away from the interface when the pD is below 5, and point towards the surface when the pD is higher than 5, with strong hydrogen bonds towards the surface.},
keywords = {P11, pre-TACO},
pubstate = {published},
tppubtype = {article}
}
Diebold, Ulrike; Rupprechter, Günther
Preface: Surface Science of functional oxides
Journal ArticleIn: Surface Science, vol. 681, pp. A1, 2019.
Links | BibTeX | Tags: P02, P08, pre-TACO
@article{Diebold2019,
title = {Preface: Surface Science of functional oxides},
author = {Ulrike Diebold and Günther Rupprechter},
doi = {10.1016/j.susc.2018.11.017},
year = {2019},
date = {2019-03-01},
journal = {Surface Science},
volume = {681},
pages = {A1},
publisher = {Elsevier BV},
keywords = {P02, P08, pre-TACO},
pubstate = {published},
tppubtype = {article}
}

Singraber, Andreas; Behler, Jörg; Dellago, Christoph
Library-Based LAMMPS Implementation of High-Dimensional Neural Network Potentials
Journal ArticleIn: Journal of Chemical Theory and Computation, vol. 15, no. 3, pp. 1827–1840, 2019.
Abstract | Links | BibTeX | Tags: P12, pre-TACO
@article{Singraber2019a,
title = {Library-Based LAMMPS Implementation of High-Dimensional Neural Network Potentials},
author = {Andreas Singraber and Jörg Behler and Christoph Dellago},
doi = {10.1021/acs.jctc.8b00770},
year = {2019},
date = {2019-01-24},
journal = {Journal of Chemical Theory and Computation},
volume = {15},
number = {3},
pages = {1827--1840},
publisher = {American Chemical Society (ACS)},
abstract = {Neural networks and other machine learning approaches have been successfully used to accurately represent atomic interaction potentials derived from computationally demanding electronic structure calculations. Due to their low computational cost, such representations open the possibility for large scale reactive molecular dynamics simulations of processes with bonding situations that cannot be described accurately with traditional empirical force fields. Here, we present a library of functions developed for the implementation of neural network potentials. Written in C++, this library incorporates several strategies resulting in a very high efficiency of neural network potential-energy and force evaluations. Based on this library, we have developed an implementation of the neural network potential within the molecular dynamics package LAMMPS and demonstrate its performance using liquid water as a test system.},
keywords = {P12, pre-TACO},
pubstate = {published},
tppubtype = {article}
}
Reticcioli, Michele; Sokolović, Igor; Schmid, Michael; Diebold, Ulrike; Setvin, Martin; Franchini, Cesare
Interplay between Adsorbates and Polarons: CO on Rutile TiO2(110)
Journal ArticleIn: Physical Review Letters, vol. 122, no. 1, pp. 016805, 2019.
Abstract | Links | BibTeX | Tags: P02, P07, pre-TACO
@article{Reticcioli2019,
title = {Interplay between Adsorbates and Polarons: CO on Rutile TiO_{2}(110)},
author = {Michele Reticcioli and Igor Sokolović and Michael Schmid and Ulrike Diebold and Martin Setvin and Cesare Franchini},
doi = {10.1103/physrevlett.122.016805},
year = {2019},
date = {2019-01-09},
journal = {Physical Review Letters},
volume = {122},
number = {1},
pages = {016805},
publisher = {American Physical Society (APS)},
abstract = {Polaron formation plays a major role in determining the structural, electrical, and chemical properties of ionic crystals. Using a combination of first-principles calculations, scanning tunneling microscopy, and atomic force microscopy, we analyze the interaction of polarons with CO molecules adsorbed on the reduced rutile TiO_{2}(110) surface. Adsorbed CO shows attractive coupling with polarons in the surface layer, and repulsive interaction with polarons in the subsurface layer. As a result, CO adsorption depends on the reduction state of the sample. For slightly reduced surfaces, many adsorption configurations with comparable adsorption energies exist and polarons reside in the subsurface layer. At strongly reduced surfaces, two adsorption configurations dominate: either inside an oxygen vacancy, or at surface Ti_{5c} sites, coupled with a surface polaron. Similar conclusions are predicted for TiO_{2}(110) surfaces containing near-surface Ti interstitials. These results show that polarons are of primary importance for understanding the performance of polar semiconductors and transition metal oxides in catalysis and energy-related applications.},
keywords = {P02, P07, pre-TACO},
pubstate = {published},
tppubtype = {article}
}

Cheng, Bingqing; Engel, Edgar A; Behler, Jörg; Dellago, Christoph; Ceriotti, Michele
Ab initio thermodynamics of liquid and solid water
Journal ArticleOpen AccessIn: Proceedings of the National Academy of Sciences, vol. 116, no. 4, pp. 1110–1115, 2019.
Abstract | Links | BibTeX | Tags: P12, pre-TACO
@article{Cheng2019,
title = {Ab initio thermodynamics of liquid and solid water},
author = {Bingqing Cheng and Edgar A Engel and Jörg Behler and Christoph Dellago and Michele Ceriotti},
doi = {10.1073/pnas.1815117116},
year = {2019},
date = {2019-01-04},
urldate = {2019-01-04},
journal = {Proceedings of the National Academy of Sciences},
volume = {116},
number = {4},
pages = {1110--1115},
publisher = {Proceedings of the National Academy of Sciences},
abstract = {A central goal of computational physics and chemistry is to predict material properties by using first-principles methods based on the fundamental laws of quantum mechanics. However, the high computational costs of these methods typically prevent rigorous predictions of macroscopic quantities at finite temperatures, such as heat capacity, density, and chemical potential. Here, we enable such predictions by marrying advanced free-energy methods with data-driven machine-learning interatomic potentials. We show that, for the ubiquitous and technologically essential system of water, a first-principles thermodynamic description not only leads to excellent agreement with experiments, but also reveals the crucial role of nuclear quantum fluctuations in modulating the thermodynamic stabilities of different phases of water.},
keywords = {P12, pre-TACO},
pubstate = {published},
tppubtype = {article}
}
